MAVIS Hub Edition 117
Published 3 February 2021
1. Product News
Availability of Lactating Cow Intramammary Antibiotics
Carprieve 5% w/v Small Animal Solution for Injection - Product defect recall alert
2. Enforcement
Animal medicines seizure notice: Border Force, Heathrow Airport (1)
Animal medicines seizure notice: Border Force, East Midlands Airport
Animal medicines seizure notice: Border Force, Heathrow Airport (2)
Animal medicine seizure notice: Parcel addressed to County Tyrone
Animal medicine seizure notice: Parcel addressed to Bordon, Hampshire
Animal medicine seizure notice: Parcel addressed to Coalisland, County Tyrone (1 of 2)
Animal medicine seizure notice: Parcel addressed to Coalisland, County Tyrone (2 of 2)
Animal medicine seizure notice: Parcel addressed to Strabane, County Tyrone
Animal medicine seizure notice: Parcel addressed to Castlederg, County Tyrone
Animal medicine seizure notice: Parcel addressed to Banbridge, County Down
3. AMR (Antimicrobial Resistance)
3.1 Policy
Priorities for this year include continued implementation of animal health-specific aspects of the cross-government UK AMR National Action Plan (2019-2024) and production of a Case Study report detailing the UK’s reduction in sales and use of antibiotics for food-producing animals.
The report will be published by the Food and Agriculture Organisation of the United Nations and highlight the value of the multi-sectoral voluntary collaborative approach adopted in the UK.
3.2 Defra Antimicrobial Resistance Co-ordination (DARC)
The DARC group met virtually on 24th November 2020 to discuss the recent trends in antibiotic resistance (AMR) in bacteria of importance to animal and public health.
The meeting included updates from the Veterinary Medicines Directorate and Animal and Plant Health Agency, including a presentation on the key findings from the recently published UK-VARSS report.
Minutes from previous meetings are available on the DARC group webpage on GOV.UK
4. Inspections
We have published an online register of sites authorised to manufacture veterinary medicines including, where applicable, their good manufacturing practice (GMP) certificates.
5. Guidance Updates
Bee medicines: availability in the UK
Centrally Authorised Products conversion explainer
From 1 January 2021 Application and Authorisation Process explainer
From 1 January 2021 Immunological Batch Release explainer
From 1 January Veterinary Medicines Regulations explainer
Controlled drugs: recording, using, storing and disposal
From 1 January 2021 changes to the cascade explainer
From 1 January 2021 Maximum Residue Limits explainer
Veterinary medicines unfettered access from Northern Ireland
Submission of an application for an animal medicine authorisation
Apply for a Marketing Authorisation in the UK for a veterinary medicine or expire one
Marketing authorisations for veterinary medicines
Apply for batch release of an immunological veterinary medicine
Variations to a veterinary Marketing Authorisation or Homeopathic Remedy
Authorisations to manufacture veterinary medicines
Pharmacovigilance of Veterinary Medicines in Great Britain
Common Statement Canada-United Kingdom (as of 1 January 2021)
From 1 January 2021 Import and export of veterinary medicines explainer
From 1 January 2021 MAH and Authorised Personnel Location
Fees applied to animal medicine authorisation applications
Veterinary medicine wholesale dealer’s authorisation (WDA)
The cascade: prescribing unauthorised medicines
United Kingdom-Canada Regulatory Cooperation: Guidance on Veterinary Drug Simultaneous Reviews
Veterinary Pharmacovigilance responsibilities for authorisation holders
Report a product defect: veterinary medicine
Apply to renew a marketing authorisation for a veterinary medicine
6. Research and statistics
Residues of veterinary medicines in food: 2020
UK veterinary antibiotics sales among lowest levels recorded in Europe
Veterinary Medicines Pharmacovigilance Annual Review 2019 Summary
VMD Published Standards 2020 to 2021: Monitoring performance
Top ten imported veterinary medicines - Quarterly report 1 October 2020 to 31 December 2020
7. Stakeholder Engagement
Joint VMD and VPC open information day - 18 November (updated)
VMD Regulatory Science Strategy – we would like your views
Reminder about sending post to the VMD
United Kingdom - Canada Regulatory Cooperation
8. Corporate News
Defra: gender pay gap report and data 2020
Recruitment of Non-Executive Director
FOI requests received by the VMD between 1 July to 31 December 2019
9. Organogram as at 31 January 2021
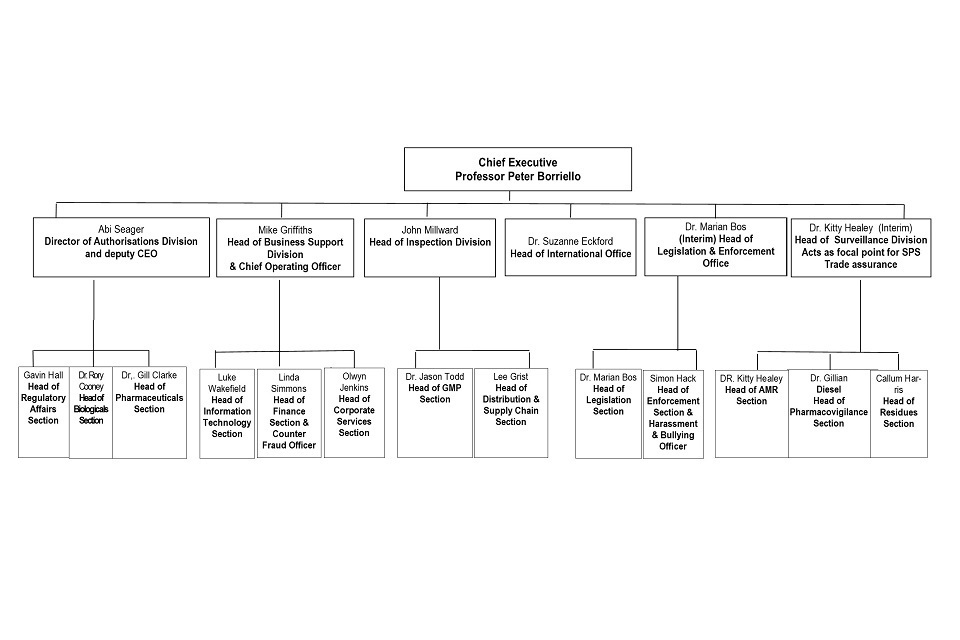
Organogram
