HPR volume 8 issue 41: news
Updated 23 December 2014
1. New vaccine coverage data updates for pertussis and rotavirus programmes published
The latest vaccine coverage estimates for the pertussis in pregnancy vaccination programme, up to August 2014, and for the childhood rotavirus vaccination programme, up to September 2014, are published in the Infection Reports section of this issue of the HPR [1,2].
The pertussis in pregnancy vaccine coverage report includes new monthly estimates of the proportion of women vaccinated during pregnancy who gave birth in the period April through to August 2014, and compares these data with estimates from the beginning of 2014. Average coverage between April and August 2014 was around 54%, a decline on the 60% reported earlier in the year, but coverage appears to be gradually increasing.
The rotavirus vaccine coverage report shows that high coverage was rapidly achieved for the first cohorts of children offered this vaccine routinely in England: 92.6% of the children evaluated at 25 weeks of age between February and September 2014 had received the first rotavirus vaccine dose and 87.5% had completed the two-dose course, according to data from 90% of all GP practices in England.
1.1 References
-
“Pertussis Vaccination Programme for Pregnant Women: vaccine coverage estimates in England, April to August 2014, HPR 8(41): infection reports, 24 October 2014.
-
“National rotavirus immunisation programme: preliminary data for England, October 2013 to September 2014”, HPR 8(41): infection reports, 24 October 2014.
2. Increase in endemic Meningococcal group W (MenW) ST-11 complex associated with severe invasive disease in England and Wales
England and Wales is experiencing an upsurge in invasive capsular group W (MenW) disease. Historically, MenW incidence has been low, accounting for only 1-2% of invasive meningococcal disease (IMD) cases annually. An increase in 2000-2002 was associated with pilgrimage to Hajj. After the introduction of a mandatory meningococcal vaccination for pilgrims, MenW cases in England and Wales declined to pre-2000 levels. Since 2009/10, MenW cases have increased year-on-year such that there were 98 confirmed cases during June 2013 to July 2014 (2013/14), accounting for 15% of all invasive meningococcal cases. This increase has been observed across all English regions. Phenotypic characterisation of the MenW isolates revealed that the increase was nearly all caused by MenW strains phenotypically expressing PorB serotype 2a, associated with the cc11 clonal complex.
MenW:2a initially increased in older adults but, by 2012/13, had extended across all age groups. Epidemiological follow-up of cases did not identify any association with travel or recent entry into the UK, nor was there any evidence of clustering of cases within households. Detailed clinical follow-up of laboratory-confirmed MenW cases diagnosed during 2010/11-2012/13 did not identify any differences in MenW:2a and non-2a infections in terms of clinical presentation, disease severity or case fatality rate. However, for the first time since the Hajj outbreak, there were several deaths associated with MenW:2a in infants, young children, adolescents and young adults during 2012/13 and 2013/14. Prior to this period, MenW-associated deaths occurred almost exclusively among ≥45 year-olds.
Since the Hajj-associated international outbreaks, a number of countries in Latin America, sub-Saharan Africa, South Africa and the Far East have reported increases in endemic MenW disease caused by strains belonging to cc11. In Europe, some countries such as France have observed an increase in MenW cases that were linked with travel to – or contact with someone who had travelled to – sub-Saharan Africa where MenW is epidemic.
So far, however, no European country other than the UK has reported an increase in endemic MenW disease. The emergence and rapid clonal expansion with continuing increase in endemic MenW:cc11 disease in the UK is concerning, particularly because natural immunity against this capsular group is low across all age-groups. MenW is potentially preventable through vaccination with the quadrivalent MenACWY conjugate vaccine and, if this trend continues, we may need to consider modifying the current immunisation programme to offer additional protection against MenW.
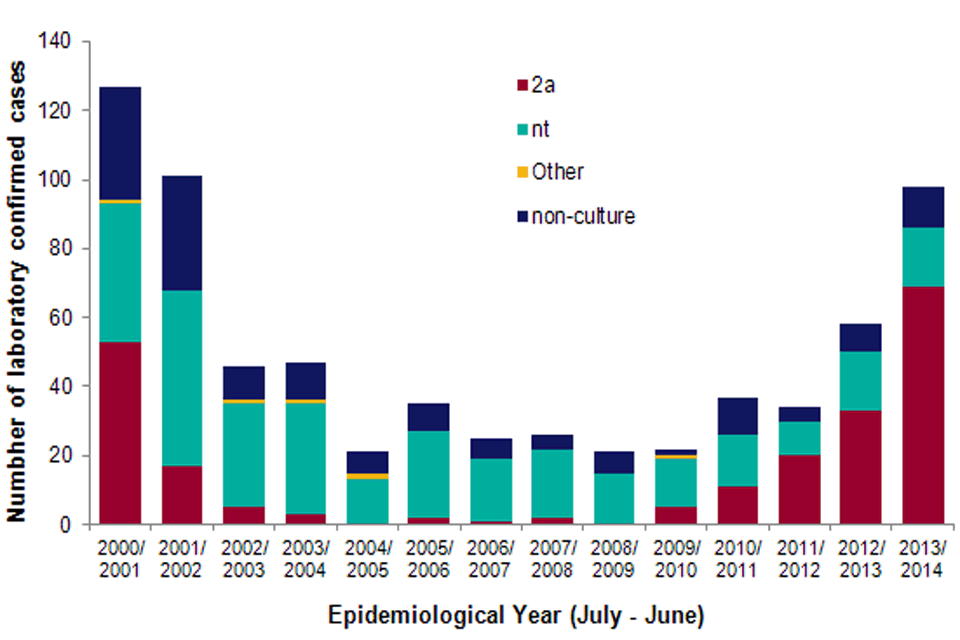
Laboratory-confirmed cases of invasive meningococcal disease group W, by type, during the 2000/01-2013/14 epidemiological years (provisional data).
3. National Poisons Information Service annual report 2013/14
The PHE-commissioned National Poisons Information Service (NPIS) has published its latest annual report, for 2013/14 [1]. NPIS is a clinical toxicology service for health care professionals working in the NHS with operational units in Birmingham, Cardiff, Newcastle and Edinburgh that provide expert advice on all aspects of acute and chronic poisoning, supporting best practice in the diagnosis and management of patients who may have been accidentally or deliberately poisoned, whether by ingestion, injection, inhalation or skin or eye contact.
NPIS encourages NHS staff seeking advice on the diagnosis, treatment and care of patients who may have been poisoned to use its online database, TOXBASE, as a first point of call for information, with its telephone enquiry service being devoted to the more complex cases. The most frequent TOXBASE users are hospital departments and NHS 111, NHS 24 and NHS Direct staff. The annual report is a statement of activity, accountability and governance and includes workload statistics, and the results of quality assurance exercises and of surveillance activities.
Online enquiries (“user sessions”) on TOXBASE continued the steady rise of recent years, totalling more than 576,000 during the period covered by the new report – an increase of circa 14% compared to the previous year. There was a 3.5% increase in telephone enquiries (including calls to the UK Teratology Information Service), to a total 55,669.
Areas of particular interest in 2013/14 included: drugs of misuse, including newer psychoactive substances; paracetamol; 2,4-dinitrophenol; radiation; pesticides; reed diffusers; and electronic cigarettes.
On drugs of misuse, as in its 2012/13 report, the NPIS notes a significant increase in enquiries related to novel psychoactive substances (so-called ‘legal highs’). This year enquiries about synthetic cannabis substitutes (synthetic cannabinoid receptor agonists, SCRAs) have increased 13-fold, so these now represent the second most common drug of misuse encountered in NPIS telephone enquiries after cocaine. TOXBASE accesses for SCRAs also more than doubled over the previous year, putting them among the top 10 drugs of misuse accessed [2].
Since 2012, NPIS has established a system of enhanced surveillance for “chemicals of interest for toxicosurveillance”, including chlorine, carbon monoxide, ammonia and hydrogen cyanide. Carbon monoxide was the most frequent subject of both TOXBASE accesses (more than a quarter of accesses) and case enquiries followed up by NPIS staff in 2013/14. The next most frequently accessed TOXBASE entries were those for chlorine, hydrogen cyanide, ammonia, hydrofluoric acid, paraquat, formaldehyde, CS gas, propane and alkalis, respectively.
NPIS is commissioned by PHE through its Centre for Radiation, Chemical and Environmental Hazards.
3.1 References
- National Poisons Information Service annual report 2013/2014 (October 2014), ISBN 978-0-85951-761-4. Downloadable from the NPIS website.
- “Legal high’ and e-cigarette poison queries climb”, PHE press release, 23 October 2014.
4. European syndromic surveillance guidelines published
European guidelines on syndromic surveillance in human and animal health – aimed at increasing European capacity for real-time surveillance and monitoring of infectious disease outbeaks and other threats to public health or safety – have been published by the “Triple-S” project (Syndromic Surveillance Survey, Assessment toward Guidelines for Europe) [1,2].
Started in 2010, the European Commission-funded project involved 24 organisations from 13 European countries, including PHE’s Real-time Syndromic Surveillance Team, who are leading exponents of the discipline [3].
An early output of Triple-S was the development of a common European definition of syndromic surveillance [4], helping to improve understanding and to move towards harmonisation of national approaches. An inventory of European syndromic surveillance identified 33 systems in 15 countries concerned with human health, and 27 systems in 12 countries concerned with animal health.
The European guidelines, the main output of Triple-S, are intended for public health professionals and epidemiologists who would like to use or develop syndromic surveillance to support existing surveillance systems and public health monitoring. A final output of the project is a European strategy for syndromic surveillance which offers a number of proposed models for enhancing the use and integration of syndromic surveillance to support public health surveillance in Europe.
All outputs from Triple-S are available from the project website [2].
4.1 References
- Hulth A (2014). First European guidelines on syndromic surveillance in human and animal health published, Euro Surveill. 2014; 19(41).
- “Triple-S – the syndromic surveillance project”.
- Public Health England. “Syndromic Surveillance Systems and Analyses”.
- “Triple S. Assessment of syndromic surveillance in Europe”, Lancet. 2011; 378(9806): 1833-4.
