HPR volume 14 issue 6 (24 and 25 March)
Updated 23 December 2020
COVID-19 guidance and microbiology: fourth update
Issuance of updated social distancing guidance and stay at home advice for the general public, and detailed advice on shielding extremely vulnerable people, were significant additions to GOV.UK guidance following the closure of businesses and other venues in the UK from 21 March 2020. These new priorities in the government’s response to COVID-19 were reflected in a restructuring of the main GOV.UK landing page (gov.uk/coronavirus), now titled ‘Coronavirus (COVID-19): what you need to do’.
Earlier, the move from the containment to the delay phase of the government response, on 16 March, followed recognition of the fact that imported cases were a reducing proportion of total UK confirmed cases and that the principal threat was from secondary and sporadic UK-acquired cases. The lessening significance of imported cases, which became apparent in mid-March, is illustrated by the (partial) epidemic curve for the UK, covering late February and early March, shown below, produced using data derived from the intensively-investigated, so-called “First Few Hundred” UK cases.
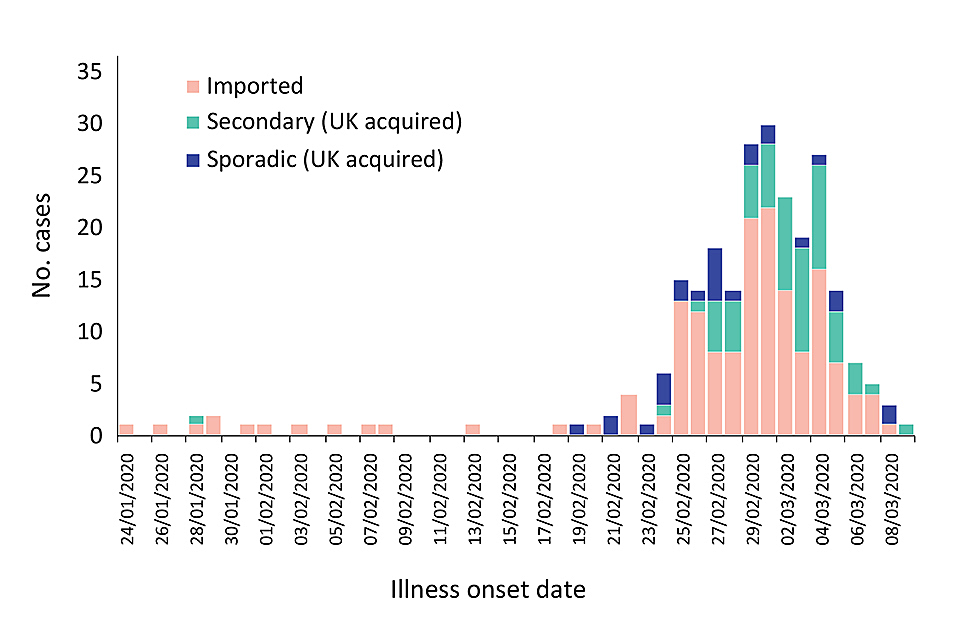
Sample of UK confirmed cases, as at 11 March, and epidemic curve.
Whereas in the early stages of the UK epidemic (during the containment phase), UK cases were almost exclusively imported, “sporadic” cases began to be recorded from the end of February and sustained secondary transmission became the predominant cause of infection from early March.
New guidance
Significant targeted guidance issued during the last fortnight has been primarily for non-clinical settings, including for:
- vulnerable children and young people
- schools and other education settings
- prisons and other prescribed places of detention
- transport hubs including airports and train stations
- freight transport
- hostels and day centres for people sleeping rough
Expansion of testing
A robust, specific RT-PCR diagnostic test for COVID-19 was developed and validated by PHE’s National Infection Service central laboratories in Colindale in early January. More than 30,000 tests had been delivered to the healthcare system by Public Health England (PHE) and NHS laboratories by early March. The scale of testing activity is to be multiplied in the coming weeks, with some capacity also being provided by commercial providers.
Study of the clinical virology of the infection, gained from a study of the first UK patients via PHE’s HCID network, has been complemented by on-going sentinel surveillance organised through the RCGP network of corresponding medical practices (involved in routine seasonal influenza surveillance).
While PHE Colindale retains a primary testing-service delivery capacity for coronavirus surveillance in London, where the majority of UK cases continue to occur, the capability of PHE regional laboratories and NHS laboratories to use National Infection Service (NIS) diagnostics continues to be steadily expanded.
In addition, 2 further strands of COVID-19-related microbiology activity are being rapidly pursued: sentinel surveillance testing for the virus in the general UK population (to complement data derived from symptomatic and/or suspect cases); and the development of serology tests to further inform the UK epidemic response.
During a recent internal presentation to PHE staff, Maria Zambon, NIS Director of Reference Microbiology (and virology lead in the NIS’s COVID-19 incident response arrangements) paid tribute to her colleagues in the NIS molecular diagnostics team who had developed the PCR test chosen to be rolled out nationally. “The scientific impact of work in public health laboratories is not always recognised,” Zambon said. But, in this case, the significant contribution made by those who developed the diagnostics rolled out nationally, on which the country relies for its response, was clear. Zambon also praised the serology and virus isolation team’s work that had been crucial in the development of diagnostics and serology tests.
Infection reports in this issue of HPR
Group A streptococcal infections: second report on seasonal activity in England, 2019/20
Tetanus in England: 2019
