Common animal-associated infections in England: 2025
Published 26 March 2026
Applies to England
Introduction
The common animal-associated infections annual report is produced by the Emerging Infections and Zoonoses team in the Travel Health, Zoonosis, Emerging Infections, Respiratory & TB Division (TARZET), Epidemic and Emerging Infections Directorate, UK Health Security Agency (UKHSA). As of 2023, the data presented in these reports is for England only. Data for Wales is reported separately by Public Health Wales. Previous reports presenting data for both England and Wales are available,
This report summarises laboratory diagnosed cases of selected zoonotic infections reported in England from 1 January to 31 December 2025 through the Second Generation Surveillance System (SGSS) and/or pathogen specific reference laboratories. It is important to note that not all cases of a zoonotic infection will present to healthcare services, with some cases treated empirically based on clinical suspicion without the need for a confirmatory test. Therefore, the numbers in this report are expected to be an underestimate of the true burden of zoonotic disease in England, likely biased towards severe infections that are more likely to present to healthcare. Furthermore, not all zoonoses presented in this report are notifiable in England. As such, it is likely not all confirmed laboratory test results are reported to UKHSA. For a list of notifiable diseases and causative organisms in England please see the Notifiable diseases and causative organisms: how to report.
This report is produced using a finalised dataset and thus data presented here is not ‘provisional’ as in previously published quarterly reports. However, changes can still occur to numbers reported due to late notifications and updates to records as more information becomes available. Therefore, data presented in the most recent report supersedes those previously published in quarterly reports.
The report includes descriptive analysis on the trends, age and sex distribution, geographical distribution and where available, exposure types. Rates of infections were calculated using population denominators for the respective year and geography derived from the Office of National Statistics (ONS). At the time of publishing, 2025 population data was not available, therefore population denominators for 2024 were used as a proxy for calculating rates of infection.
Regional data is presented by UKHSA regions (East Midlands, East of England, London, North East, North West, South East, South West, West Midlands, and Yorkshire and The Humber) and Integrated Care Boards (ICBs) for mapping. ICBs cover a specific geographical area and are NHS organisations responsible for planning health services for their local population. Geographical mapping at ICB level provides more granular insights on geographical spread which may be masked by higher levels such as UKHSA region. Rates at ICB level refer to the population served by the ICB. Regional data is based on the location of the patient’s residential postcode where available. If patient postcode is not available, then location of the referring hospital or laboratory is used. Therefore, regional data presented does not necessarily reflect the geographic area where the cases are located or where the infection may have been acquired. Where relevant and available, travel history has been included, however this does not necessarily reflect where the infection was acquired.
The impact of the public health measures implemented in England due to the COVID-19 pandemic during 2020 and 2021 should be considered when making comparisons with this time period.
Q-fever data for 2025 is not presented in this annual report. As part of an internal review, the data is being validated and will be published when available. Lyme disease will now be reported as part of the annual vector borne disease report, due for publication in April 2026.
Main points
Key findings from this report are that:
- Of the infections presented in this report, Hepatitis E virus (HEV) followed by Pasteurella spp. represent the greatest number of cases.
- Confirmed cases of brucellosis have decreased compared with the previous year, from 31 cases, in 2024, to 23 cases in 2025.
-
Cases of Hepatitis E virus (HEV) have been steadily increasing annually since 2017 to 2025, exceeding 2017 levels by 102.6% (from 735 to 1489 cases), despite a decline during the COVID-19 pandemic. Investigations are ongoing to better understand what is driving these increases:
- between 2024 and 2025, cases increased by 2.4% from 1452 to 1489 cases
- sharp increases of 14.9% were noted in rates of incidence in females between the ages of 15 and 44 between 2024 and 2025, from 1.95 to 2.24 cases per 100,000 population, respectively
- The number of leptospirosis cases decreased slightly in 2025 (115 cases) compared to 2024 (156 cases), however, the rate per 100,000 population has remained relatively consistent (0.27 in 2024 to 0.20 per 100,000 population in 2025).
- Psittacosis case numbers remained broadly stable in 2025 (13 cases) compared to 2024 (11 cases). We note an increase in samples referred for testing for Chlamydia psittaci and/or Chlamydia abortus at the UKHSA Respiratory and Vaccine Preventable Bacteria Reference Unit (RVPBRU) in 2025, however positivity rates remained relatively stable when compared with previous years.
- There was a 12.9% increase in toxoplasmosis cases reported in 2025 (228 cases) compared with 2024 (202 cases). Overall trends have been broadly stable with some fluctuations between 2017 to 2025.
- Reports of toxigenic Corynebacterium ulcerans slightly increased from 2024 to 2025, from 10 to 13 cases.
- Reports of Pasteurella species have been increasing annually since 2017 (with the exception 2020, coinciding with the COVID-19 pandemic). There was an 8.9% increase in cases from 924 to 1007 from 2024 to 2025, respectively.
- Other less common zoonotic pathogens reported include Capnocytophaga species, Mycobacterium marinum, Erysipelothrix rhusiopathiae, and Taenia species, saw varying fluctuations. Number of cases are small for these pathogens and so should be interpreted with caution.
Trends in England
Overview
Of the infections presented in this report, we note the greatest number of cases reported for hepatitis E virus (HEV) followed by Pasteurella species (spp.). We also note an incremental trend in both from 2020, following a slight decline in 2020, coinciding with the COVID-19 pandemic (Figure 1a). Subtle increases in case numbers in recent years are also notable for Mycobacterium marinum, taeniasis, brucellosis, toxigenic Corynebacterium ulcerans and Erysipelothrix rusiopathiae, although overall case numbers are small (Figure 1b). Other zoonotic pathogens remain broadly stable with some fluctuations, although numbers are small for some pathogens (Figure 1a and b).
Figure 1. Animal associated infections in England: annual total laboratory reports by pathogen, 2017 to 2025**
(a) Pathogens with counts greater than 100; (b) Pathogens with counts fewer than 100

* The diagnostic tests for leptospirosis changed in August 2020, therefore data is presented from 2021 onwards
** Data reported in figures 1a and 1b reflects total laboratory confirmed and probable case numbers of animal associated infections. References may be made in this report to confirmed and probable case numbers, reported separately in-text.
Brucellosis caused by Brucella species
Brucella bacteria can be rough or smooth strain - this refers to the appearance of the bacteria when grown on an agar plate. Smooth strains including Brucella melitensis (B. melitensis), Brucella abortus (B. abortus) or Brucella suis (B. suis) are typically more pathogenic in humans than rough strains such as Brucella canis (B. canis) and are the primary cause of brucellosis in humans.
Brucellosis is rare in England and human cases are usually caused by smooth strains of Brucella species often acquired following consumption of unpasteurised dairy products in countries where brucellosis is endemic. The UK is officially free of brucellosis in cattle, sheep and goats since 1985. The Brucella Reference Unit (BRU) is a specialist laboratory that provides diagnostic, clinical management, and reference services for Brucella spp. and is the primary hub for human Brucella diagnostics in the UK. The BRU provide serology testing for smooth strain species and a pan-Brucella species PCR test, which is only accredited for smooth strains of Brucella spp. Only cases tested and reported to the BRU are included in this section. The following case definition has been applied for brucellosis surveillance:
Confirmed smooth strain brucellosis is any person with:
- isolation of smooth strain Brucella spp. from any clinical site OR;
- a titre of 1:160 or higher on standard agglutination testing.
B. canis is a recognised zoonotic pathogen affecting dogs and is not considered endemic in the UK dog population. Since 2020, there has been an increase in the number of reports of B. canis infection in dogs, the majority of which have been in dogs imported into the UK from Eastern Europe (1). Sporadic human cases are reported globally, but there is no routine surveillance data available for comparison with other countries. In 2022, the UK diagnosed the first confirmed human case of brucellosis caused by B. canis, linked to an imported dog from Eastern Europe (2).
In contrast to smooth strain Brucella species (B. melitensis, B. abortus or B. suis), there are currently no specific accredited serological or molecular tests for B. canis in humans. Definitive diagnosis of B. canis in humans is through culture only. In 2023, the Brucella Reference Unit (BRU) adopted canine serology tests (B. canis serum agglutination test (SAT) and a canine B. canis IgG ELISA test), alongside the pan-Brucella species PCR test to support clinical diagnosis. Following a review of PCR test results in 2024, the following case definitions have been applied for brucellosis surveillance in England from 2022 onwards:
Confirmed Brucella canis is any person with isolation (culture) of Brucella canis from a clinical specimen.
Probable Brucella canis is any person with a clinically compatible illness with:
- B. canis specific antibody response (B. canis IgG ELISA or Serum Agglutination Test) with seroconversion or increase in titres on convalescent/paired serology testing OR;
- Other B. canis specific antibody response AND detection of human pathogenic Brucella spp. nucleic acid in clinical specimen AND negative non-canis serology.
Data for laboratory confirmed brucellosis cases was obtained from the BRU, reported through the Second Generation Surveillance System (SGSS).
Epidemiological analysis of brucellosis
In 2025, there were 23 reported cases of brucellosis, of which, 22 were identified as smooth strain Brucella species (B. melitensis, B. abortus or B. suis), and 1 probable B. canis. No confirmed cases of B. canis were reported in 2025 (Figure 2). There were fewer brucellosis cases reported in 2025 (23 cases) compared with 2024 (31 cases). We note a peak in case numbers in 2024, which is potentially linked to overseas travel returning to pre-pandemic levels, however we note cases are greater than pre-pandemic and 2025 levels. The number of samples tested by the BRU has also been increasing annually since 2021. The reduction in B. canis cases may be linked to a change in public health guidance, as testing is now only advised for exposed individuals with symptoms of brucellosis.
Figure 2. Brucellosis cases in England, 2017 to 2025*
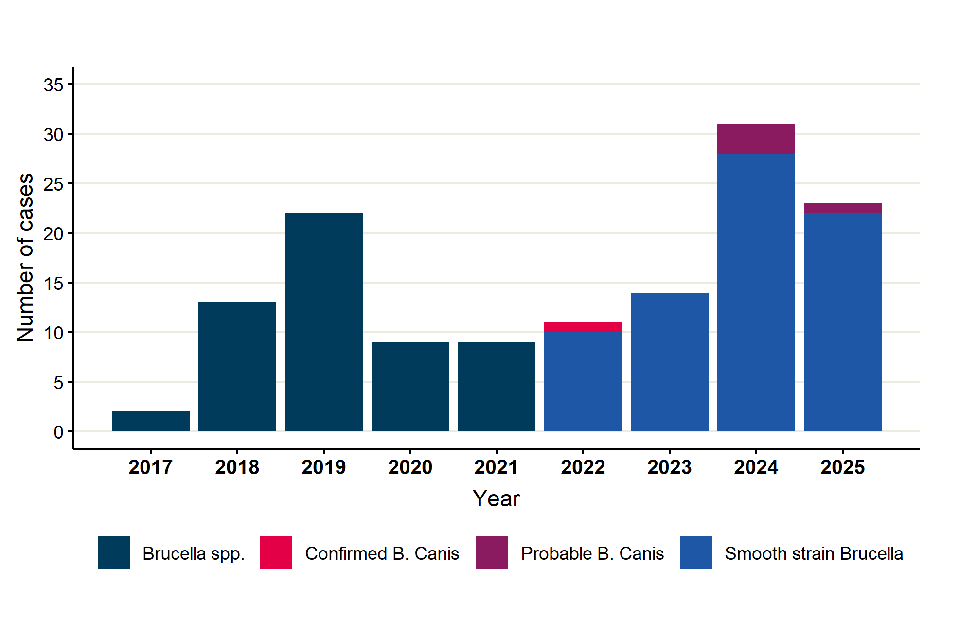
- For cases prior to 2022, the results are not speciated and any person with a positive laboratory result is included as a brucellosis case. Smooth strain Brucella species are B. melitensis, B. abortus or B. suis. Serological testing for smooth strain Brucella species has been included for surveillance purposes from 2019.
Of the 22 smooth strain brucellosis cases reported in 2025, 6 (27.3%) were female, 13 (59.1%) were male and 3 (13.6%) did not have sex recorded (Figure 3). This is a shift from the more even sex distribution of cases observed in 2024, but in keeping with the ECDC who reported that 65% of brucellosis cases were male (3). The median age of cases was 52 years (range 16 to 83 years), which is in line with what has been seen previously.
Figure 3. Percentage of smooth strain brucellosis cases in England by age group and sex, 2025*

- This excludes 3 cases with unknown age or sex.
Based on patient postcode, the regions with the highest number of cases in 2025 were London (5 cases) and the North West (4 cases).
For cases where exposures were recorded, 15 (68.2%) reported travel to countries where brucellosis is endemic, including Iraq (3 cases), Somalia (3 cases), India (2 cases), Ethiopia, Israel, Jordan, Kenya, Qatar, Turkey and Europe (1 case each). The proportion of cases who had travelled to Western Asia and Eastern Africa has remained consistent over the past 2 years (Figure 4). Of the cases who reported travel to countries where brucellosis is endemic, 6 reported consuming unpasteurised dairy products during their visit. There were 2 cases of chronic brucellosis, both males aged 75-84 years, with known occupational exposure to cattle and sheep in the UK, prior to Great Britain being officially brucellosis free in 1985. No travel information was available for these cases. For the remaining 5 (22.7%) cases, there was no travel history exposure recorded.
There was 1 probable B. canis case reported in 2025, a female in the 15 to 44 years age group. The case experienced symptoms consistent with acute brucellosis and had potential travel and occupational exposures. Due to changes in testing associated with B. canis in 2023, it is not possible to draw direct comparisons between current data and data prior to 2023.
Figure 4. Smooth strain brucellosis cases in England by region of travel, 2024 to 2025*

*Travel information is not available for cases prior to 2024. Unknown means that there was no travel history exposure recorded. Smooth strain Brucella species are B. melitensis, B. abortus or B. suis. World regions of travel were assigned based on the United Nations world region classifications (4).
Hepatitis E virus (HEV)
Hepatitis E virus (HEV) is the most common cause of acute viral hepatitis in England, which can cause inflammation of the liver. Infection usually occurs through the faecal-oral route in humans, but there is evidence that HEV infection can be acquired from food products, namely pork products (5). There are 8 different HEV genotypes, 5 of which cause disease in humans. Genotype 1 (G1) and G2 viruses are predominantly found in low- and middle-income countries and are transmitted through the faecal oral route, whereas G3, G4, and G7 viruses are responsible for infections in high-income countries and are primarily foodborne (6). In the UK HEV G3 group 2 is the predominant cause of infections, these infections are not associated with travel but are likely to be associated with consuming pork products from outside the UK (5,6).
In 2025, a review of surveillance case definitions for HEV was conducted in conjunction with the UKHSA Blood Borne Virus Unit Colindale. Due to the changes to surveillance case definitions, the number of confirmed acute hepatitis E cases reported for 2017 to 2025 have changed compared with previous reports. Data reported here supersedes those previously published. The following updated case definitions were used to analyse data:
Confirmed acute infection:
A case is considered confirmed acute when the results clearly indicate a recent or active HEV infection. This includes: (i) any reactive PCR result alone, OR (ii) patterns where IgM is reactive, with or without IgG.
Chronic Infection:
A chronic HEV infection is defined as a PCR reactive result three months or longer from the earliest positive PCR test indicating persistent viral replication.
Data for HEV includes laboratory data from the Public Health Laboratory Birmingham and the UKHSA Blood Borne Virus Unit Colindale reference laboratories, and through the Second Generation Surveillance System (SGSS). There has been a shift in testing and reporting of HEV cases over time, with increased testing and reporting of HEV from local laboratories as testing capabilities improve. The two reference laboratories now predominantly provide reference testing as opposed to primary diagnostics. Therefore, the combined data set provides a comprehensive reflection of the number of acute HEV cases reported in England.
These data do not include cases identified through screening of blood donations by NHS Blood and Transplant (NHSBT); the annual number of donations in which HEV is identified and any transmission events can be found in the NHSBT safe supplies annual report (7). Data from SGSS are taken as laboratory confirmed acute infection, with the above definitions not applied due to limitations in available data.
Data was de-duplicated across all datasets for cases using a 90-day episode window (3 months), utilising the earliest specimen date and strongest result (confirmed over probable), and only confirmed cases were reported for surveillance in this report. Any cases that had recurring reactive PCR tests following the 90-day episode window were defined as chronic cases.
Epidemiological analysis of Hepatitis E virus (HEV)
There were 1,489 laboratory confirmed acute cases of HEV infection reported in 2025, an increase of 2.5% compared to 2024 (1,452 cases), with the rate of infection also increasing from 2.50 in 2024 to 2.57 cases per 100,000 population in 2025 (Figure 5). Further, there were 135 chronic cases of hepatitis E identified between 2017 to 2025.
Numbers of HEV cases, and rate of infection has increased annually since 2017, with a notable decline in 2020, followed by annual increases. Between 2017 and 2025 cases and rate of infection have increased by 102.6% (735 cases in 2017 to 1,489 cases in 2025), and 91.8% (1.34 cases per 100,000 population in 2017 to 2.57 cases per 100,000 population in 2025), respectively (Table 1). Investigations are ongoing to better understand these increases.There were no outbreaks of HEV reported in 2025.
Figure 5. Counts and rates per 100,000 population of cases of laboratory confirmed acute hepatitis E infection in England by year, 2017 to 2025 (95% CI)

Table 1: Annual rates per 100,000 population of cases of laboratory confirmed acute hepatitis E infection in England, 2017 to 2025 (95% CI)
| Year | Number of cases | Population | Rate per 100,000 population | 95% Confidence Interval (CI) |
|---|---|---|---|---|
| 2017 | 735 | 54,971,772 | 1.34 | 1.24 - 1.44 |
| 2018 | 844 | 55,293,743 | 1.53 | 1.43 - 1.63 |
| 2019 | 1,094 | 55,615,291 | 1.97 | 1.85 - 2.09 |
| 2020 | 886 | 55,726,600 | 1.59 | 1.49 - 1.70 |
| 2021 | 1,081 | 55,975,530 | 1.93 | 1.82 - 2.05 |
| 2022 | 1,331 | 56,548,317 | 2.35 | 2.23 - 2.48 |
| 2023 | 1,365 | 57,355,631 | 2.38 | 2.26 - 2.51 |
| 2024 | 1,452 | 58,046,289 | 2.50 | 2.37 - 2.63 |
| 2025 | 1,489 | 58,046,289 | 2.57 | 2.44 - 2.70 |
Of the 1,489 HEV cases reported in 2025, 795 (53.4%) were male (age range from 0 to 96 years, median 58 years), 692 (46.4%) were female (age range from 3 to 96 years, median 52 years) and 2 (0.2%) cases where sex was unknown (Figure 6). There is a persisting predominance of HEV in middle aged and older males (aged 45 years and over), with 585 (39.2%) total cases in 2025 in this category, with the highest case counts in males aged 45 to 64 years, 303 cases (20.3%), followed by females 15 to 44 years, (259 cases (17.4%) (figure 6). However, in 2025 there was a notable increase in female cases across several age groups, particularly the 15 to 44 age category with 259 (17.4%) cases in 2025 in this group, compared with 225 (15.5%) cases in 2024. Females aged over 85 also had an increase in case numbers, with 36 cases (2.4%) in 2025 compared with 30 cases (2.1%) in 2024.
Figure 6. Percentage of laboratory confirmed acute cases of hepatitis E infection in England by age group and sex, 2025*

- This excludes 2 cases where sex was unknown
There was an increase in the rate of infection in females in these age groups as well. The rate of infection in females aged 15-44 increased from 1.94 cases per 100,000 population (95% CI 1.70 to 2.21) in 2024 to 2.24 cases per 100,000 population (95% CI 1.97 to 2.53) in 2025, and the rate of infection in females aged above 85 increased from 3.28 cases per 100,000 population (95% CI 2.21 to 4.68) to 3.94 cases per 100,000 population (95% CI 2.76 to 5.45). Rates of HEV infection increased in older age groups (75+ years old) for both males and females in 2025. Rates of HEV infection in individuals (both male and female) aged 75+ spiked in 2022, with a decline until 2024, before rising again in 2025 (Figure 7). There is an ongoing further investigation of these trends.
Overall rates of HEV infection were generally higher in males (2.79 cases per 100,000 population (95%CI 2.61 to 2.99) than females (2.34 cases per 100,000 population in females (95% CI 2.17 to 2.52) in 2025, aligning with data from previous years. This finding is consistent with previous surveillance studies in HEV infection in England and Wales, which found that non-travel associated cases were higher in older men infected with HEV (genotype 3) (5). Whilst information on exposures is not routinely gathered for cases, previous investigations have identified associations with pork and pork related products in non-travel related cases (5,6).
Figure 7. Annual rates per 100,000 population of cases of laboratory confirmed acute hepatitis E infection in England by age group and sex, 2017 to 2025

At a regional level, highest rates of HEV infection in 2025 were seen in the West Midlands, and the South East regions, with rates of infection of 3.3 cases per 100,000 population (95% CI: 2.9 to 3.8) and 3.1 cases per 100,000 population (95% CI: 2.7 to 3.4), respectively. These regions have seen the greatest rates of infection for the past five years. London and the North West also saw high rates of infection intermittently over the last five years, with both regions experiencing steady increases in case rates since 2023. The North East experienced a 44.4% increase in HEV infection rate from 2024 to 2025, from 1.8 cases per 100,000 population (95% CI: 1.4 to 2.4) in 2024 to 2.6 cases per 100,000 population (95% CI: 2.0 to 3.2) in 2025. The South West region has experienced a consistent decline in rates of infection from a peak of 4.6 cases per 100,000 population (95% CI: 4.1 to 5.2) in 2022 to 2.8 cases per 100,000 population (95% CI: 2.4 to 3.3) in 2025 (Figure 8).
There is an ongoing investigation into the increase in rates of HEV infection in 2022 in specific regions, particularly in individuals aged 75 years and older.
Figure 8. Annual rates per 100,000 population of cases of laboratory confirmed acute hepatitis E infection in England by UKHSA region, 2017 to 2025.

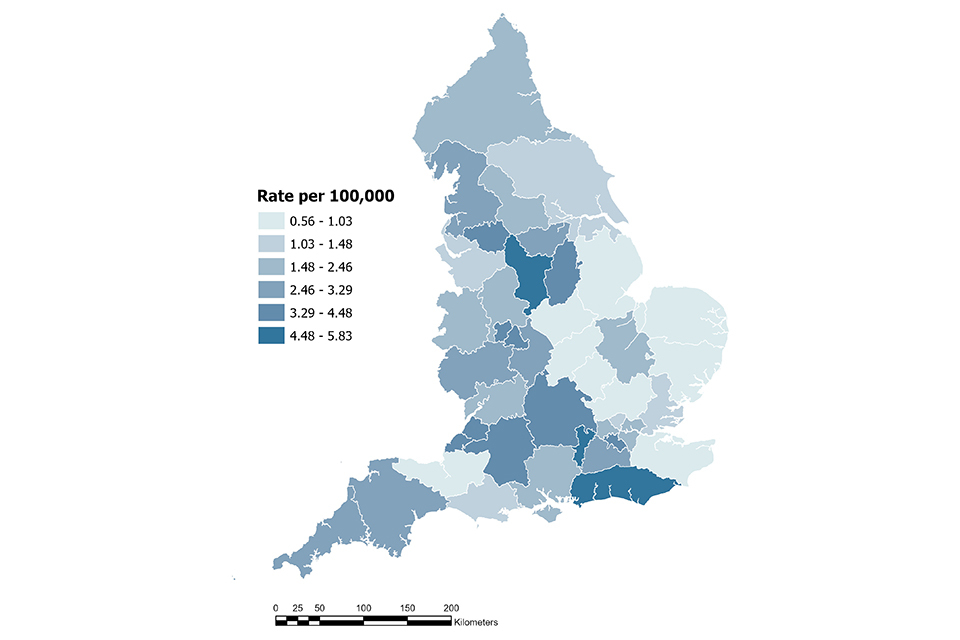
Rates of HEV infection vary geographically. In 2025 the highest rates were observed in the following ICB regions: NHS Derby and Derbyshire ICB (5.84 cases per 100,000 population; 95% CI: 4.49 to 7.45), NHS Sussex ICB (5.40 cases per 100,000 population; 95% CI: 4.37 to 6.60), and NHS Frimley ICB (4.73 cases per 100,000 population; 95% CI: 3.35 to 6.49). Lowest rates of HEV infection were reported from NHS Norfolk and Waveney ICB (0.57 cases per 100,000 population; 95% CI: 0.21 to 1.23) (Figure 9). These data are based on the patient’s postcode where available. Where a patient’s postcode is unavailable, the location of the referring hospital or laboratory is used.
Figure 9. Rates per 100,000 population of cases of laboratory confirmed acute hepatitis E infection in 2025, by ICB.
Leptospirosis
Leptospirosis is a bacterial infection caused by Leptospira genus, most commonly L. interrogans and related strains. Wild and domestic animals can spread the infection through their urine, most commonly rodents, cattle and pigs. Transmission to humans can occur through direct exposure to animal urine, or exposure to urine-contaminated environments such as canals, rivers or lakes, through the skin or mucous membranes. Infection is particularly common among agricultural workers who are more likely to be exposed to infected rodents, livestock, and water sources. Leptospirosis cases are reported worldwide but are more prevalent in tropical regions.
Data on leptospirosis testing were obtained from the Laboratory Information Management System used by the Rare and Imported Pathogens Laboratory (RIPL, UKHSA Porton).
As the assays used for leptospirosis diagnosis changed in August 2020, data are presented here from 2021 onwards (8).
Confirmed case: as of 1 August 2020, a laboratory confirmed case of leptospirosis is defined by a positive 16S rRNA PCR result only
Probable case: A probable case is defined by a strongly positive immunoglobulin M (IgM) result and compatible clinical history without a confirmatory PCR result. Probable cases are usually treated clinically on the basis of this result. National surveillance of probable cases was introduced in 2021.
Epidemiological analysis of leptospirosis
In 2025, the rate of infection for both confirmed and probable leptospirosis cases was 0.20 per 100,000 population in England (115 cases; 95% CI: 0.16 to 0.24). This is a small decrease in the number of cases compared to 2024, however the rate has remained relatively consistent (156 cases; 0.27 per 100,000 population; 95% CI: 0.23 to 0.31 in 2024) (Figure 10, Table 2).
Of the 115 overall cases reported in 2025, 63 (54.8%) were confirmed and 52 (45.2%) were probable cases. This represents an infection rate of 0.11 confirmed cases per 100,000 population in 2025 (95% CI: 0.08 to 0.14), a decrease from 2024 (102 confirmed cases, 0.18 per 100,000, 95% CI: 0.14 to 0.21). Conversely, the number of probable cases remained similar when comparing the data from 2024 (54 probable cases) and 2025 (Figure 10). There was no change to the specific diagnostic assays used or reporting from 2024 to 2025. There was also no large difference in the number of tests performed. Therefore, this may represent a true decrease in confirmed leptospirosis cases in 2025.
It should be noted that, due to changes in assigning surveillance definitions for leptospirosis, the number of probable cases reported here for 2021 and 2022 are lower than in previous reports. Data reported here supersedes those previously published.
Figure 10. Counts and rates per 100,000 population of leptospirosis cases (confirmed and probable), in England by year, 2021 to 2025 (95% CI)

Table 2: Annual rates per 100,000 population of leptospirosis cases (confirmed and probable) in England, 2021 to 2025 (95% CI)
| Year | Cases | Population | Rate per 100,000 population | 95% Confidence Interval (CI) |
|---|---|---|---|---|
| 2021 | 99 | 55,975,530 | 0.18 | 0.14 - 0.22 |
| 2022 | 99 | 56,548,317 | 0.18 | 0.14 - 0.21 |
| 2023 | 139 | 57,355,631 | 0.24 | 0.2 - 0.29 |
| 2024 | 156 | 58,046,289 | 0.27 | 0.23 - 0.31 |
| 2025 | 115 | 58,046,289 | 0.20 | 0.16 - 0.24 |
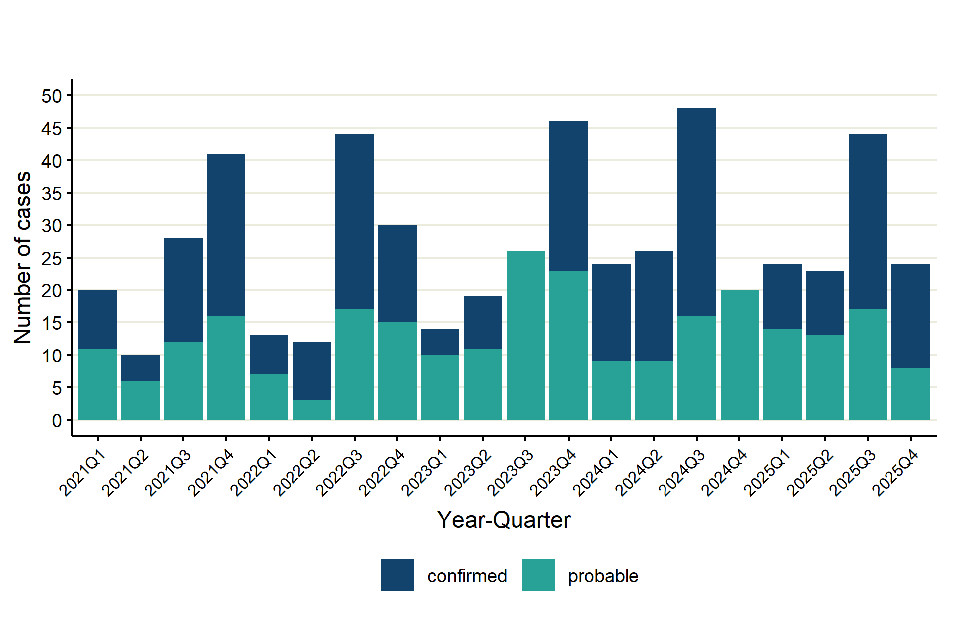
Greater numbers of leptospirosis cases are observed in the third and fourth quarter each year, indicating seasonality of cases in summer and autumn months, coinciding with greater travel during this period (Figure 11).
Figure 11. Number of leptospirosis cases (confirmed and probable) in England by quarter, Q1 2021 to Q4 2025
Of the 115 cases in 2025, 83 (72.2%) were male (age range from 4 to 71 years, median 41 years) and 31 (27.0%) were female (age range from 9 to 76 years, median 44 years). There was one (0.9%) case where sex was unknown.
The greatest proportion of cases was seen in the 15 to 44 years age group for both males and females, with a particular skew to this age group in males (42.1%) compared to females (14.0%) (Figure 12).
Sex distribution of leptospirosis cases has been consistent across years – of the 493 cases (confirmed and probable) reported between 2021 and 2024, 80.8% were male. The consistently higher incidence of leptospirosis infection in males over several years in England aligns with global surveillance studies (9,10).
The age distribution may indicate that these age groups are more likely than others to have occupational exposure to the urine of infected animals through agriculture work or lifestyle exposures such as recreational freshwater sports.
Figure 12. Percentage of leptospirosis cases (confirmed and probable) in England by age group and sex, 2025*

*This excludes one case where sex was unknown
Based on patient postcode, the UKHSA regions with the highest number of cases in 2025 were London (28 cases) and the South West (22 cases). Between 2024 and 2025, rates of leptospirosis per 100,000 population decreased or remained stable in all regions except London, where rates have increased since 2023, from 0.16 per 100,000 population (95% CI: 0.09 to 0.26) to 0.31 per 100,000 population (95% CI: 0.21 to 0.45) in 2025 (Figure 13).
Figure 13. Annual rates per 100,000 population of leptospirosis cases (confirmed and probable) in England, by UKHSA region, 2021 to 2025

Further granulation of geographical data at ICB level for 2025, show rates of leptospirosis were greatest in the following ICBs (0.51 to 0.59 cases per 100,000): NHS Bristol, North Somerset and South Gloucestershire, NHS Devon, NHS Cornwall and the Isles of Scilly, NHS Somerset, and NHS Lincolnshire (Figure 14). Where patient postcode was not available, the location of the referring hospital or laboratory was used. Location of cases does not necessarily reflect the geographic area where the infection was acquired.
Figure 14. Rates per 100,000 population of leptospirosis cases (confirmed and probable) in 2025, by ICB

Of the 115 leptospirosis cases (confirmed and probable) in 2025, information on recent travel abroad (in the last 30 days) was available for 58 (50.4%) cases. However, travel history is not always available, and the reported country of recent travel may not necessarily reflect where the infection was acquired. With this caveat in mind, almost half (57/115, 49.6%) of cases in 2025 reported recent travel abroad.
Of the 57 cases who reported travel abroad, the most common region reported was Latin America and the Caribbean (18 cases). Cases also reported travel to South-eastern Asia (13 cases), Southern Asia (13 cases), Europe (8 cases), and Central Asia, Eastern Africa, Eastern Asia and North America (1 case each) (Figure 15).
Figure 15. Leptospirosis cases in England by region of recent travel, 2021 to 2025*

*Cases who reported travel to multiple regions are counted in each region. World regions of travel were assigned based on the United Nations world region classifications (4).
Exposure information was available for 32 (27.8%) of the 115 leptospirosis cases in 2025. However, as exposure information is not always available, the numbers reported here are likely an underestimate. In addition, it is important to bear in mind that these exposures may have been in the UK or abroad.
Of the 32 cases with exposure information, 24 (75.0%) reported potential exposure to a water source, with activities including swimming (12 cases), fishing (4 cases), kayaking (1 cases), white-water rafting (1 cases) in freshwater, and the remaining water source exposures being unspecified (6 cases). Ten cases (31.3%) reported contact with a rodent or rodent excreta as a potential exposure. Two of these cases reported both rodent and water contact as potential exposures.
Pateurellosis
Pasteurellosis is an infection caused by Pasteurella species, typicallty transmitted by animal bites, scratches or licks on broken skin. Animals do not need be clinically unwell to transmit the organism, as carriage can occur in the absence of symptoms. Pasteurella multocida is the most common cause of infection, often following a bite or scratch from domestic pets.
A new surveillance definition for pasteurellosis was implemented in 2025. There is limited information available on surveillance definitions for Pasteurella species, from available literature published by the World Organisation for Animal Health (WOAH), invasive infections were defined as Pasteurella spp. isolated from a usually sterile site, which is corroborated by a study in the USA (11,12). Using this, we have defined pasteurellosis in the context of surveillance as follows:
Confirmed case: Definitive laboratory confirmation; isolation of Pasteurella spp. from a normally sterile site
Probable case: Definitive laboratory confirmation; isolation of Pasteurella spp. from a non-sterile site, where Pasteurella is a recognised pathogen
Possible case: Definitive laboratory confirmation; isolation of Pasteurella spp. from a non-sterile site, where colonisation is a possibility
Data was de-duplicated at patient level and a 6-week rolling episode window (42 days) was applied to repeat infections, retaining the earliest specimen date and most severe outcome (severe being confirmed followed by probable and possible) or result for each episode. Limited published guidance exists on the definition of episode windows for pasteurellosis; only one study, conducted in Australia, has previously defined such windows, classifying all subsequent isolations of the same species within 30 days as representing a single episode (13). Given the nature of treatment pathways and the characteristics of laboratory data held within the Second Generation Surveillance System (SGSS), a 42-day episode window was considered more appropriate for the purposes of this analysis.
Epidemiological analysis of pasteurellosis
In 2025, a total of 1007 cases of pasteurellosis were reported in England, representing an increase of 8.9% when compared with 924 cases reported in 2024. Species-level characterisation was available for the majority of cases, with Pasteurella multocida accounting for 648 (64.3%) cases and Pasteurella canis for 186 (18.5%) cases. The overall rate of infection in 2025 was 1.73 per 100,000 population (95% CI: 1.63 to 1.85), compared with 1.59 per 100,000 population (95% CI: 1.49 to 1.70) in 2024, indicating a modest but upward trend in incidence (Table 3, Figure 16). It is important to note that although the incidence rate of Pasteurella has increased annually since 2020, the same is not true for confirmed cases in isolation, which fluctuate but remain at a relatively constant rate between 0.2 and 0.3 per 100,000 of the population.
Figure 16. Counts and rates per 100,000 population of pasteurellosis cases in England by year, 2017 to 2025 (95% CI)

Table 3: Annual rates per 100,000 population of all pasteurellosis cases (confirmed, probable, and possible) in England, 2017 to 2025 (95% CI)
| Year | Cases | Population | Rate per 100,000 population | 95% Confidence Interval (CI) |
|---|---|---|---|---|
| 2017 | 673 | 54,971,772 | 1.22 | 1.13 - 1.32 |
| 2018 | 709 | 55,293,743 | 1.28 | 1.19 - 1.38 |
| 2019 | 752 | 55,615,291 | 1.35 | 1.26 - 1.45 |
| 2020 | 659 | 55,726,600 | 1.18 | 1.09 - 1.28 |
| 2021 | 756 | 55,975,530 | 1.35 | 1.26 - 1.45 |
| 2022 | 841 | 56,548,317 | 1.49 | 1.39 - 1.59 |
| 2023 | 873 | 57,355,631 | 1.52 | 1.42 - 1.63 |
| 2024 | 924 | 58,046,289 | 1.59 | 1.49 - 1.70 |
| 2025 | 1007 | 58,046,289 | 1.73 | 1.63 - 1.85 |
Of the 1007 2025 cases, 559 were female, with a rate of 1.88 (95% CI: 1.72 to 2.04) per 100,000 population, whilst 434 were male with a rate of 1.51 (95% CI: 1.37 to 1.66) per 100,000 population, and 14 were unknown sex (Figure 17).
Figure 17. Percentage of all pasteurellosis cases (confirmed, probable, and possible) in England by age group and sex, 2025*

*This excludes 14 cases with unknown sex
At a regional level, rates of confirmed cases fluctuate annually, with some notable exceptions. East of England has seen a consistent rise in cases from 2021 onwards, with the greatest increase between 2024 (27 confirmed cases) and 2025 (40 confirmed cases). Yorkshire and the Humber has also seen a consistent increase since 2022, rising from 123 to 159 cases in 2025. Further investigation is ongoing to understand these increases (Figure 18).
Figure 18. Annual rates per 100,000 population of confirmed pasteurellosis cases in England by UKHSA region, 2017 to 2025

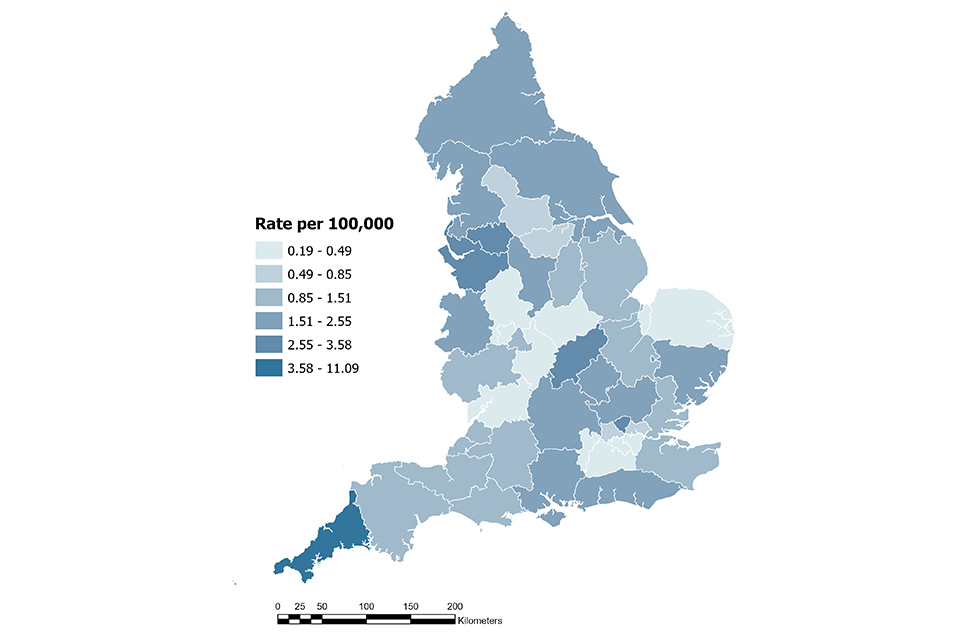
At an ICB level we note higher rates of infection in Greater Manchester ICB at 3.6 cases per 100,000 population (95% CI: 2.9 to 4.3), Northamptonshire ICB at 3.5 cases per 100,000 population (95% CI: 2.3 to 5.1) and Chesire and Merseyside ICB at 3.0 cases per 100,000 population (95% CI: 2.4 to 3.7), Cornwall and the Isles of Scilly ICB rate of 11.09 cases per 100,000 population (95% CI: 8.5 to 14.1).
Figure 19. Rates per 100,000 population of all pasteurellosis cases in 2025, by ICB
Chlamydia psittaci / Chlamydia abortus
Psittacosis is a bacterial infection in humans caused by Chlamydia psittaci, which can cause respiratory illness and pneumonia. Most common routes of transmission are inhalation of infected respiratory particles, or dust contaminated with faeces from pet birds such as parrots, parakeets, budgerigars, and cockatiels. Not all infected birds are symptomatic; however, they are still able to transmit infection.
Data for C. psittaci / C. abortus were obtained from the UKHSA Respiratory and Vaccine Preventable Bacteria Reference Unit (RVPBRU) in Colindale and from cases reported by local laboratories through SGSS. It is recognised that it is difficult to diagnose infection retrospectively through serological assays and, as such, following an external review, the surveillance case definitions for psittacosis were updated in 2023. As of 1 January 2023, the following case definition has been applied for psittacosis surveillance data:
Confirmed case: A positive 16S rRNA PCR result or culture isolation only.
Serological testing is no longer routinely carried out for C. psittaci and therefore less severe cases may not be identified through laboratory testing. This case definition has been applied to all retrospective data presented in this report.
It should be noted that PCR testing is unable to differentiate between C. psittaci and C. abortus. There are currently no other reliable methods to specifically identify C. psittaci as the causative agent of a patient with suspected psittacosis disease. As such, the numbers presented in this report will reflect laboratory confirmed cases with a positive PCR result for C. psittaci / C. abortus.
Epidemiological analysis of Chlamydia psittaci / Chlamydia abortus
There were 13 laboratory confirmed cases of C. psittaci / C. abortus in 2025, representing an 8% increase compared with 2024 (11 cases). The fourth quarter of 2025 had the highest number of cases (7 cases) since the third quarter of 2023, and the highest to date since 2017, representing a 333% increase from 3 cases reported in 2017 to 13 cases reported in 2025. Overall case numbers remain low, and prior to 2022 have remained below 10 cases; however, since 2023 we note a sharper increase and annual increases, with numbers exceeding 10 cases from 2024. There were no laboratory confirmed cases of C. psittaci / C. abortus identified in England in 2022 (Figure 20). Overall counts are small and therefore data should be interpreted with caution.
Exposure information was available for 11 cases in 2025, with 9 (69.2%) cases reporting exposure to birds, 2 (15.4%) cases reporting no discernible exposure risks from birds, and 2 (15.4%) cases where exposure to birds was unknown. Of the 9 cases who reported exposure to birds; 5 cases were household exposures through owning pet birds or being in contact with a household pet bird, 3 cases reported feeding birds in public, and 1 case reported occupational exposure to birds or bird droppings.
Between 2019 and 2021 the RVPBRU noted a decrease in requests for testing likely due to the COVID-19 pandemic with sample numbers received increasing from 2022 onwards and peaking in 2025. The RVPBRU reported 216 requests for tests for C. psittaci/ C. abortus compared with 181 requests for test in 2024, an increase of 19.3%, however, positivity rates of samples remaining consistent at 4.6% (compared to 4.4% in 2024) for samples tested at the RVPBRU (positivity data are only available for samples sent to the RVPBPU). The increase in testing could be attributed to increased clinician awareness in England following a temporary increase in human cases of psittacosis in Northern Europe, reported by the WHO in March 2024 (14). A UKHSA briefing note was also circulated to clinicians in March 2024, flagging the increase in Europe, along with testing services offered by UKHSA.
Figure 20. Number of laboratory confirmed cases of C. psittaci / C. abortus in England by year, 2017 to 2025
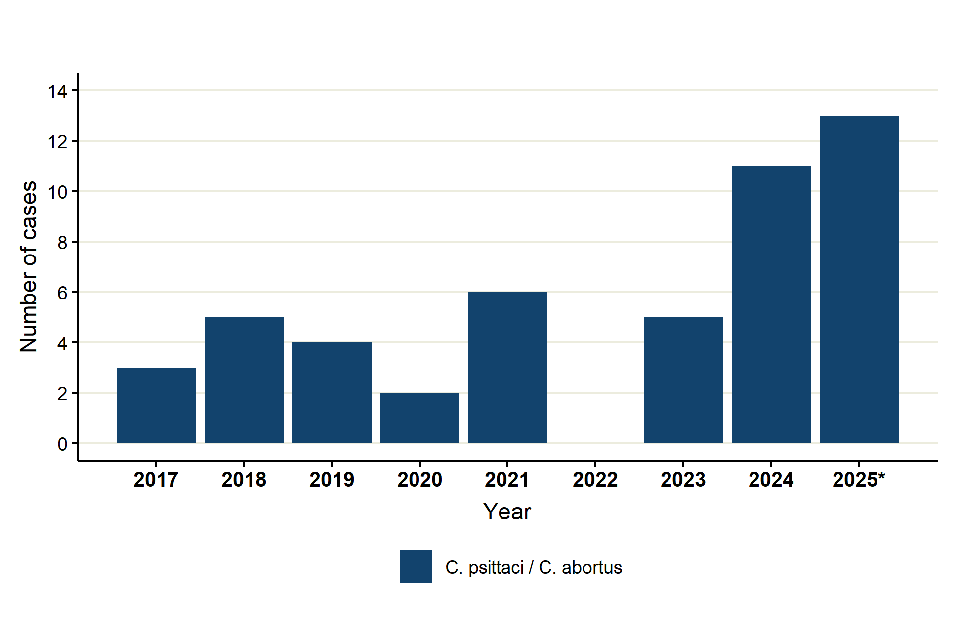
*Includes 10 cases derived from UKHSA RVPBRU reference laboratory, and 3 cases derived from SGSS
Of the 13 cases reported in 2025, 10 (76.9%) were male (age range 50 to 83 years, median age 65), and 3 (23.1%) cases were female (age range 54 to 92 years, median age 83). Most cases (6 cases, 46.2%) fell within the 45-64 age category. This contrasts data reported in 2024, where 3 cases (27.3%) were 65 and above in age. Further, males in this age group were disproportionally affected, making up 38.5% of total cases, matching trends seen in 2024, however data should be interpreted with caution due to small numbers (Figure 21).
Figure 21. Percentage of laboratory confirmed psittacosis cases in England by age group and sex, 2025

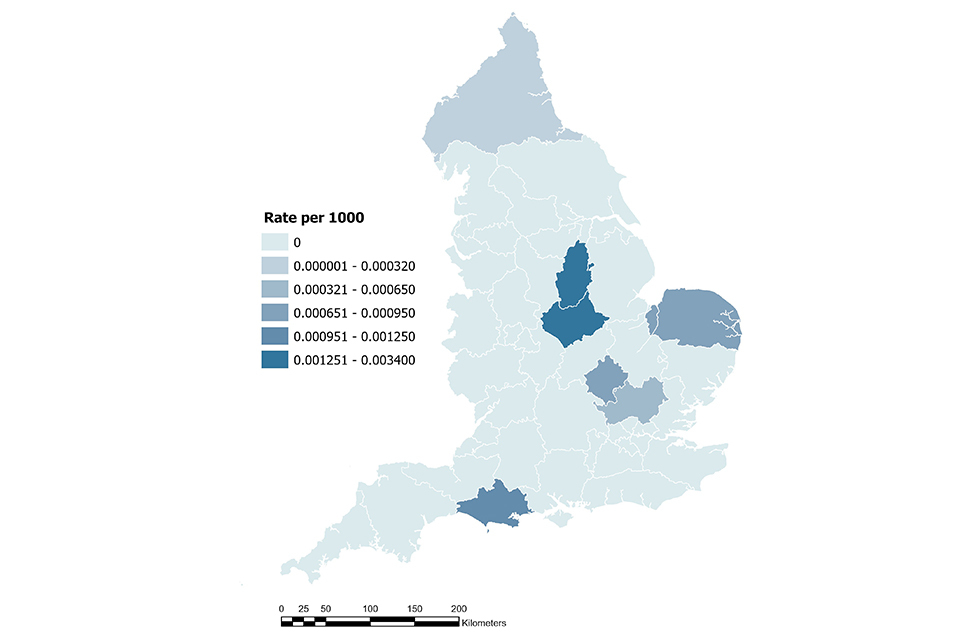
The majority of cases (8 cases, 62%) were identified in the East Midlands region, with some clustering in NHS Leicester, Leicestershire and Rutland and NHS Nottingham and Nottinghamshire ICBs, with 4 cases in each ICB, and rates of 0.003 cases per 1000 population and 0.003 cases per 1000 population, respectively (Figure 22). However, due to a small number of cases, caution must be made when interpreting rates of disease. No epidemiological links were found between cases, and further investigations are ongoing.
Three cases were identified in East of England region, one case was identified in the South West region, and one case in the North West region.
Figure 22: Rates per 1000 population (due to small case numbers) of psittacosis cases in 2025, by ICB
Toxigenic Corynebacterium ulcerans
Toxigenic Corynebacterium ulcerans (C. ulcerans), is a zoonotic pathogen that can cause respiratory or cutaneous diphtheria in humans. Further information on diphtheria cases in England can be found in the UKHSA annual diphtheria report (15).
Data on laboratory confirmed toxigenic C. ulcerans were obtained from the UKHSA Respiratory and Vaccine Preventable Bacteria Reference Unit (RVPBRU). C. ulcerans speciation is initially established by the submitting laboratory and then confirmed in RVPBRU using a combination of species-specific PCR, additional biochemical testing and MALDI-TOF MS, as required. The following definition is used for surveillance:
Confirmed toxigenic C. ulcerans case: isolation of C. ulcerans that is positive for the diphtheria toxin gene by PCR, and expresses the toxin as demonstrated using the Elek test.
Epidemiological analysis of toxigenic Corynebacterium ulcerans
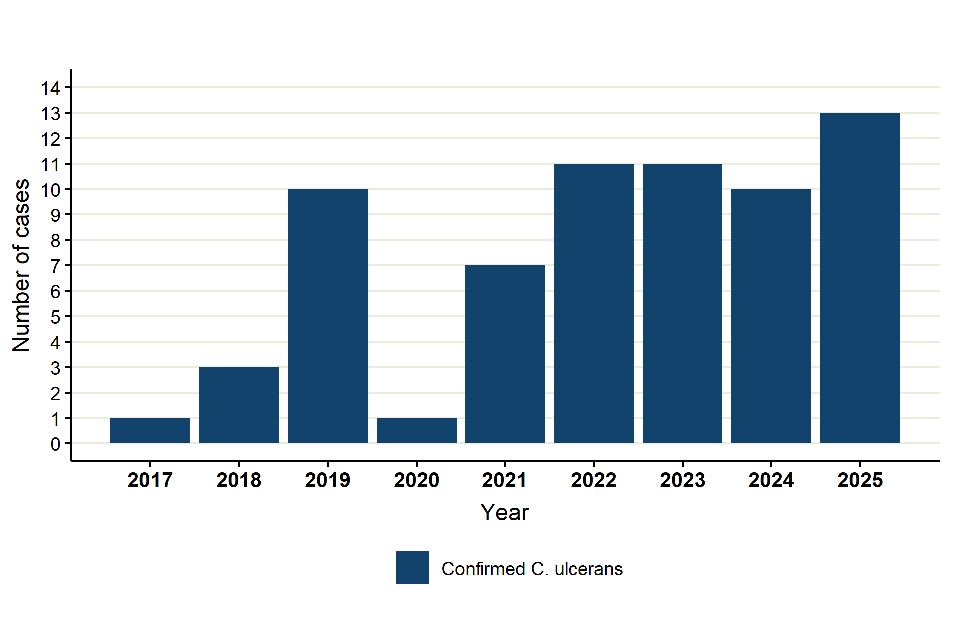
There were 13 reports of toxigenic C. ulcerans infection in 2025, an increase, of 30%, from 10 cases in 2024 (Figure 23). Case numbers rose dramatically in 2019, which was possibly an increase in ascertainment of cases, as hospitals increasingly became aware of the importance of C. ulcerans in addition to C. diphtheriae, followed by a sharp drop in 2020, coinciding with the COVID-19 pandemic. Cases have subsequently increased, with 2025 seeing the highest number of reported cases in this period.
Figure 23. Laboratory confirmed cases of toxigenic C. ulcerans in England by year, 2017 to 2025
Number of samples submitted for toxigenicity testing (at patient level) follow a similar trend to reported cases, with number of tests increasing in recent years. The overall toxigenicity rate for C. ulcerans isolated from patients was 50.0% in 2025 (13/26 cases), compared to 43.5% (10/23 cases) in 2024, and has remained broadly stable since 2023, however data should be interpreted with caution due to small numbers (Figure 24).
Figure 24. Number of toxigenic and non-toxigenic C. ulcerans isolates from humans and toxigenic positivity rate in England by year, 2017 to 2025

Of the 13 toxigenic C. ulcerans cases in 2025, 6 (46.2%) were female with a median age of 53.5 years, and 7 (53.8%) were male and had a median age of 62 years (Figure 25), which is in keeping with observations in previous years.
Figure 25. Percentage of toxigenic C. ulcerans cases in England by age group and sex, 2025

Based on patient postcode, the cases were distributed across regions, with the highest number of cases in 2025 in the East Midlands, East of England, North West, South East and Yorkshire & Humber (2 cases each). There has been no trend in geographical distribution of toxigenic C. ulcerans cases in the last few years.
Contact with animals was reported by 11 (84.6%) cases in 2025. Companion animals of 7 (63.6%) of these cases were swabbed, and 4 (36.4%) cases declined swabbing of their animals. Of those swabbed, 4 (57.1% - of 7 swabbed) had a companion animal that tested positive for toxigenic C. ulcerans. Between 2017 - 2025, 91.0% of all cases reported contact with animals. Throughout this period, 75.4% of those had at least one companion animal swabbed, and 30.4% tested positive for toxigenic C. ulcerans.
In England, contact with companion animals remains the most frequently reported exposure for individuals with confirmed toxigenic C. ulcerans infection. However, the animals may not show evidence of infection and it is not always possible to confirm the carriage of C. ulcerans in animals.
Toxoplasmosis
Toxoplasmosis is an infection caused by the Toxoplasma gondii parasite. It is a common infection spread by infected cat faeces, ingestion of infected meat or contaminated fresh produce. It is usually asymptomatic, but can cause serious illness in people who are immunocompromised. It can also be transmitted congenitally and cause serious complications to unborn babies and may cause miscarriages in pregnant women. In April 2025 congenital toxoplasmosis became a notifiable disease under the Health Protection (Notification) regulations.
Data for laboratory confirmed toxoplasmosis in England were obtained from the Toxoplasma Reference Unit (TRU, Public Health Wales, Swansea). The TRU receives only a proportion of samples for testing, which are primarily for confirmation of congenital cases or other more complex scenarios, therefore data reported is an underestimation of the true burden of disease in England.
Confirmed infections (acute and latent) are defined as testing positive using the Sabin-Feldman dye serological test that measures IgG and IgM. In-house and commercial IgM Enzyme Immunoassays and an in-house IgG avidity assay are used to confirm acute infection. An in-house PCR assay is used to test certain clinical samples to confirm active infection.
Epidemiological analysis of toxoplasmosis
228 confirmed toxoplasmosis cases were reported by the TRU in 2025. This is a 12.9% increase in the total number of confirmed cases compared with 2024 (202 cases). Confirmed cases in 2025 were higher than the five-year average from 2020 to 2024 (201 cases per year). There is no apparent seasonality identified across the five-year period (Figure 26). In 2025, toxoplasmosis had an incidence rate of 0.38 per 100,000 population (95% CI: 0.33 to 0. 44) for England. This is a 22.5% increase from 0.31 per 100,000 population (95% CI: 0.27 to 0.36) in 2024. Counts and rates have fluctuated over the years, with 2019 onwards remaining broadly stable, although a peak in 2018 is noted, which is not fully understood and no testing changes recorded.
Figure 26. Annual rates per 100,000 population of toxoplasmosis cases in England by year , 2017-2025 (95% CI)

Testing at patient level at the TRU has seen a slow decline from 2017 to 2025, with the exception of 2020 to 2022 where numbers increased before declining again. Positivity has broadly remained stable over this period, with the exception of a small spike in 2018.
Figure 27. Toxoplasmosis positivity rate and total number of samples in England per 100 samples, 2017-2025

Of the 228 TRU confirmed cases in 2025, 84 (36.8%) were male (age range 0 to 77, median 37 years, with a rate of 0.35 per 100,000 population [95% CI: 0.28 to 0.44]), 121 (53.1%) were female (age range 0 to 89, median 34 years, with a rate of 0.40 per 100,000 population [95% CI: 0.33 to 0.48]) and there were 23 cases where sex was unknown. The overall rate for all cases was 0.39 per 100,000 population (95% CI: 0.34 to 0.44). The largest number of cases were in the 25 to 44 age group (106 cases, 46.5%), with the majority of cases in this group being female (67 cases, 63.2%).
Since 2019, females have consistently made up a greater proportion of cases each year. The TRU have reported that the majority of the samples they receive for testing are cases in pregnant women and women of childbearing age. Screening for toxoplasmosis during pregnancy is not routine, however testing may be prompted based on clinical suspicion if foetal abnormalities are identified in scans, as these may result from toxoplasma infection. This is likely the reason for the larger number of female toxoplasmosis cases reported across a number of years, specifically in the 25 to 44 age group.
Figure 28. Percentage of laboratory confirmed toxoplasmosis cases of in England by age group and sex, 2025*

*Excludes 23 cases where sex is unknown.
The highest rates of infection were reported in the South East London ICB region (Figure 29), at 0.9 per 100,000 population (95% CI: 0.5 to 1.5), followed by North West London ICB at 0.7 per 100,000 population (95% CI: 0.4 - 1.1). Outside of London, Buckinghamshire, Oxfordshire and Berkshire West ICB had the next highest rate at 0.4 per 100,000 population (95% CI: 0.2 - 1.0). There is ongoing work to improve toxoplasmosis surveillance nationally between the TRU and UKHSA.
Figure 29. Rates per 100,000 population of laboratory confirmed toxoplasmosis cases, in 2025, by ICB

Other zoonotic organisms
Data for other zoonotic organisms are obtained from cases reported by local laboratories through SGSS. The region is based on the location of the patient’s postcode where available. If patient postcode is not available, then the location of the referring hospital or diagnostic laboratory is used. For these organisms, a 28-day rolling episode window was used to remove repeat infections, where the earliest case was kept for reporting.
Capnocytophaga species
Capnocytophaga species are frequently carried in the mouths of companion animals (cats and dogs) or humans and may be associated with an animal or human bite, or opportunistic infections in those with impaired immune systems.
There were 124 cases of Capnocytophaga infection in 2025, which is 28 more cases than 2024. Of the cases reported in 2025, 72 were further speciated to Capnocytophaga canimorsus (found in the oral flora of canine and feline species), 26 were Capnocytophaga sputigena (part of normal human oral flora) and 26 were Capnocytophaga other.
Figure 30. Laboratory confirmed cases of Capnocytophaga spp. in England by year, 2017 to 2025

Of those speciated to Capnocytophaga canimorsus, 45 cases were male and 27 were female, with the majority being in the 45-64 age group, and most cases were reported in the South West (17 cases).
Mycobacterium marinum
Mycobacterium marinum is a bacteria found in both salt and fresh water and can cause skin lesions in humans and fish. Humans can be exposed to Mycobacterium marinum through direct contact with contaminated water or infected fish.
There were 27 cases of Mycobacterium marinum infection in 2025, the same number of cases reported in 2024. We note an increasing trend in case numbers since 2017, with the exception of a drop between 2019 and 2022, with cases increasing 42.1% from 19 in 2017 to 27 in 2025.
Of cases reported in 2025,19 cases were male, and 8 cases were female, which is a similar proportion to previous years. For both male and female cases, the majority were aged 45-64 years old. Cases were reported in all regions, with the South West having the most (6 cases), followed by East of England (5 cases), and the South East (4 cases). There was no exposure information available for these cases.
Figure 31. Laboratory confirmed cases of Mycobacterium marinum in England by year, 2017 to 2025

Erysipelothrix rhusiopathiae
Erysipelothrix rhusiopathiae are bacteria found in various animals including pigs, turkeys and sheep. Transmission to humans usually occurs through direct contact with infected animals or their excretions. However, it is important to note that exposure data is not currently recorded for these cases.
There were 16 cases of Erysipelothrix rhusiopathiae reported in 2025, exceeding numbers reported in 2024 (12 cases). Of the cases in 2025, 10 were male and 6 female. Cases were located in the London region (4 cases), and the East of England, South East and Southwest (3 cases each). The East Midlands, North West and West Midlands each had a single case.
Figure 32. Laboratory confirmed cases of Erysipelothrix rhusiopathiae in England by year, 2017 to 2025

Taeniasis
Taeniasis is the infection of humans with the adult tapeworm of Taenia saginata, Taenia solium or Taenia. asiatica. Humans become infected by ingesting raw or undercooked infected meat from cattle (Taenia saginata) and pigs (T. solium and T. asiatica). Cases in the UK are most likely related to travel or imported meat.
There were 41 reports of taeniasis in 2025, which is similar to the number of cases in 2024 (39 cases). Of these, 11 were further speciated to T. saginata, and 3 to T. solium, with the remaining 27 cases unspecified. 23 cases of taeniasis were male, and 12 were female, with 6 being unknown.
The vast majority of all cases were in the 15-44 age group. Cases were reported in all regions, though this does not necessarily reflect where a case became infected. The East of England having the most (10 cases), followed by the North West (7 cases) and the South East and West Midlands (6 cases each).
Figure 33. Laboratory confirmed cases of Taeniasis in England by year, 2017 to 2025

References
1. UKHSA (2023). ‘Brucella canis: information for the public and dog owners’
2. Djokic V, Freddi L, De Massis F, Lahti E, Van Den Esker MH, Whatmore A, and others (2023). ‘The emergence of Brucella canis as a public health threat in Europe: what we know and what we need to learn’. Emerging Microbes and Infections: volume 12, issue 2
3. European Centre for Disease Prevention and Control (2024). ‘Brucellosis - Annual Epidemiological Report for 2022’
4. United Nations Standard country or area codes for statistical use (M49)’
5. Oeser C, Vaughan A, Said B, Ijaz S, Tedder R, Haywood B and others (2019). ‘Epidemiology of Hepatitis E in England and Wales: a 10-year retrospective surveillance study, 2008–2017’. Journal of Infectious Diseases: volume 220, issue 5, pages 802–10
6. Smith I, Said B, Vaughan A, Haywood B, Ijaz S, Reynolds C and others (2021). ‘Case–control study of risk factors for acquired hepatitis E virus infections in blood donors, United Kingdom, 2018–2019’. Emerging Infections and Disease: volume 27, issue 6, pages 1654–61
7. NHSBT and UKHSA (2025). ‘Safe Supplies 2024: Collaborating for Safety’
8. UKHSA (2024). ‘Leptospirosis’ [guidance]
9. Skufca J, and Arima Y. (2012). Sex, gender and emerging infectious disease surveillance: a leptospirosis case study. Western Pacific Surveillance and Response Journal (WPSAR): volume 3, issue 3, pages 37 to 39.
10. European Centre for Disease Prevention and Control (2024). ‘Leptospirosis - annual epidemiological report for 2022’
11. World Organisation of Animal Health (2021). ‘Pasteurella spp: Aetiology, epidemiology, diagnoisis, prevention and control’
12. Talley P, Snippes‐Vagnone P, Smith K (2016). ‘Invasive Pasteurella multocida infections – report of five cases at a Minnesota hospital, 2014. Zoonoses and Public Health: volume 63, issue 6, pages 431–5
13. Laupland KB, Stewart AG, Edwards F, Harris P, Heney C, George N and others (2022). ‘Pasteurella species bloodstream infections in Queensland, Australia, 2000–2019’. European Journlal of Clinical Microbiology and Infectious Disease: voume 41, number 4, pages 609–14
14. WHO European Region (2024). ‘Psittacosis’. [outbreak report]
15. UKHSA (2025). ‘Diphtheria in England – 2024 annual report. Health Protection Report: volume 19, number 4.
