Regulator asks those with diabetes to check insulin pumps
MHRA is asking people with a particular insulin pump to check the clock resets to the correct time and date when they change the battery or when the power is interrupted.
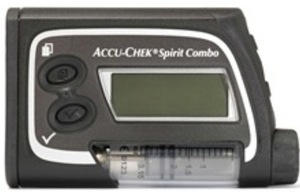
Accu-Chek Spirit Combo insulin infusion pump
A fault with the Accu-Chek Spirit Combo insulin pumps, manufactured by Roche Diagnostics Ltd, means if the power is interrupted eg during a battery change, the pump will reset to the default date and time. If this change goes unnoticed it may affect the time of insulin delivery.
John Wilkinson, MHRA’s Director of Medical Devices said:
It is vital that people check their pumps after an interruption in power supply.
People should not stop using these pumps, or switch to an alternative device, without first discussing it with a healthcare professional.
If the pump is affected by this fault, people should seek a replacement at the earliest opportunity by contacting Roche 0800 701 000.
We continue to encourage people to report any issues involving medical devices to the MHRA via our Yellow Card Scheme.
Simon O’Neill, Director of Health Intelligence at Diabetes UK said:
Ensuring the correct level on insulin is vitally important in the management of diabetes.
People using these pumps should contact the manufacturer for a replacement if they experience any problems. If they have any concerns about their level of insulin they should consult with their diabetes care team.
Background
- Please see the link to the Medical Device Alert.
- Please see link to Yellow Card Scheme.
- MHRA is responsible for regulating all medicines and medical devices in the UK by ensuring they work and are acceptably safe. All our work is underpinned by robust and fact-based judgements to ensure that the benefits justify any risks. MHRA is a centre of the Medicines and Healthcare Products Regulatory Agency which also includes the National Institute for Biological Standards and Control (NIBSC) and the Clinical Practice Research Datalink (CPRD). The MHRA is an executive agency of the Department of Health. www.mhra.gov.uk
Media enquiries
News centre
MHRA10 South Colonnade
London
E14 4PU
Email newscentre@mhra.gov.uk
Telephone (including out of hours): 020 3080 7651