Falsified Mounjaro KwikPen 15mg pre-filled pens
A falsified version of Mounjaro (tirzepatide) KwikPen 15mg solution for injection has been found supplied through one online pharmacy in the UK. The falsified product is labelled with batch D873576 and applies to Mounjaro KwikPen 15mg solution for injection in pre-filled pen only.
The issue was identified due to faults with the pens, where in all but one case the dose knob came off while in use. We have provided advice for healthcare professionals and patients as a precautionary measure who may be in possession of a 15mg pen with this batch number.
Advice for Healthcare Professionals:
- falsified Mounjaro (tirzepatide) KwikPen 15mg solution for injection in pre-filled pens have been identified. They have been supplied in the UK through one online pharmacy - The Private Pharmacy Clinic, located in Birmingham. To date 5 affected pens have been identified
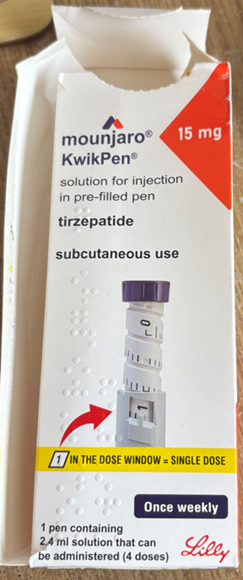
- do not supply Mounjaro KwikPen 15mg solution for injection in pre-filled pens with batch number D873576, all 15mg pens with this batch number are falsified (see pictures of product below)
- this is a legitimate batch number for genuine Mounjaro KwikPen 7.5mg, and pens at this strength are not impacted
- quarantine all remaining stock that has the affected dose and batch number and return it to your supplier
- it is likely that the substance in the pens is tirzepatide, however the pens are not of the quality and safety standards of legitimate pens and are not safe to use
- all pens identified to date have had a faulty mechanism
- if a patient reports a faulty 15mg pen, ask them to check the batch number and instruct them to discontinue use of the pen if it matches the above-mentioned batch number and contact the MHRA. Patients should always be advised not to try and extract doses from any defective pen
- the MHRA has not received any reports of patient harm that required treatment after taking this falsified medicine
- patients should be advised on signs and symptoms of allergic reactions, infection or sepsis to look out for and any treatment should be provided accordingly
- patients may require replacement medication in order to continue their course of treatment
- The General Pharmaceutical Council have served a ‘notice of conditions’ on the pharmacy involved. The MHRA is continuing with their ongoing investigations
- report suspected adverse drug reactions to tirzepatide, or suspected defective pens to the Yellow Card Scheme
Advice for Healthcare Professionals to Provide to Patients:
- a falsified version of Mounjaro (tirzepatide) KwikPen 15mg solution for injection in pre-filled pen has been identified supplied by one UK online pharmacy - The Private Pharmacy Clinic, located in Birmingham
- this pharmacy is unable to provide more of this medication at this time
- check the batch number on any Mounjaro KwikPen 15mg pre-filled pens you have been dispensed
- if you have a 15mg pen with batch number D873576, do not use the pen. Pictures of the product are provided below
- patients who have this pen should report the case to the MHRA via info@mhra.gov.uk, please include “Mounjaro Pens” in the subject line. Patients should keep the pen in a safe place, the MHRA will arrange a collection
- if you are unsure of how to identify the pen, or are unsure on whether you have administered an affected batch and have any questions please speak to a healthcare professional
- if you have administered injections using the pen already, please be reassured that, on the basis of the cases reviewed to date, the risk to you is low
- if you have administered injections, and you are experiencing any unusual symptoms that you are concerned about, seek immediate medical attention
- if you suspect that you’ve had a side effect to these affected pens, or suspect it’s not a genuine product, or have a defective pen you can report it to our Yellow Card scheme. It is important patients do not try to extract any dosage if a pen is defective in any way
- patients should be reminded on the guidance on safe use of GLP-1’s GLP-1 medicines for weight loss and diabetes: what you need to know - GOV.UK
Background
Mounjaro (tirzepatide) is a dual glucagon-like peptide-1 (GLP-1)/ glucose-dependent insulinotropic polypeptide (GIP) receptor agonist authorised for the treatment of type 2 diabetes mellitus and weight management in patients who are overweight or obese.
Eli Lilly and Company Limited has informed the MHRA that 5 falsified versions of Mounjaro (tirzepatide) KwikPen 15mg solution for injection in pre-filled pen have been identified to date, supplied by The Private Pharmacy Clinic, located in Birmingham, UK. The falsified pens are labelled as Mounjaro KwikPen 15mg solution for injection in pre-filled pen and have the batch number D873576. Please note that the expiry date of these products (10/26) is not relevant – check batch number and dose only. All pens identified to date have been faulty, often with a dose knob which comes off while in use. Eli Lilly and Company Limited have confirmed that the batch number and expiry date on the legitimate pack matches a genuine Mounjaro KwikPen 7.5mg solution for injection in pre-filled pen, whereas the falsified product packaging indicates that this product is Mounjaro KwikPen 15mg solution for injection in pre-filled pen.
Testing of the falsified pens that have been recovered confirmed that they do contain tirzepatide. However, because we do not know how the pen has been manufactured, we cannot be sure that the pen is sterile. As this is an injectable product, if its contents are not sterile, there is potential for infection. Allergic reactions may also occur. If a patient believes they have used a falsified product, they should be assessed for signs and symptoms of infection or hypersensitivity reactions.
Healthcare professionals should stop supplying the Mounjaro KwikPen 15mg pre-filled pens with the above falsified batch number immediately. Quarantine all remaining stock and return it to your supplier.
Patients who report a faulty pen should be advised not to attempt to use it. If it is a 15mg pen, they should be asked to check the batch number and report the case to the MHRA via info@mhra.gov.uk if it matches the above-mentioned batch number and expiry date. MHRA will arrange a collection of the pen.

Reporting advice
Healthcare professionals, patients, and caregivers are asked to submit reports using the Yellow Card scheme electronically using:
-
the Yellow Card app; download from the Apple App Store or Google Play Store
-
some clinical IT systems for healthcare professionals (EMIS, SystmOne, Vision, MiDatabank, and Ulysses)
When reporting suspected adverse drug reactions, please provide as much information as possible, including information about medical history, any concomitant medication, onset timing, and treatment dates.
Download document
Additional information
You can sign up to receive email notifications for Drug Safety Updates.
You can sign up to receive our monthly roundup of safety communications.
For any enquiries, please contact info@mhra.gov.uk
Article citation: MHRA Drug Safety Update volume 19, issue 7: February 2026: 3