Pregabalin 75mg capsules - incorrect strength printed in one area of foil blister pack
(Teva UK Limited) 25mg has been incorrectly printed in one position of the affected foil packs however is correct in other positions (EL(15)A/07).
12 August 2015
Class 4 medicines defect information
Caution in use
Pharmacy Level
MDR 75-07/15
Product details
Teva UK Limited
Pregabalin 75mg Capsules
PL 00289/1947
- Batch number: 018111
- Expiry date: Dec 2017
- Pack size: 1 x 56
- First distributed: 15 Jul 2015
Alert details
Teva UK Limited has notified us of a printing error on some blisters of Pregabalin 75mg capsules from the above batch. The strength is printed in several positions on the foil lid of the blisters. For affected units, the strength is printed incorrectly, as 25mg instead of 75mg, in one position on the foil. The strength is printed correctly in the remaining positions on the foil. See photograph below. The strength is stated correctly on carton labels and in the patient information leaflet. Approximately 1 in 3 blisters are affected by this issue, hence the majority of cartons from this batch are likely to contain 1 or 2 affected blisters
To ensure continuity of supply, affected stock will not be recalled. No further stock with this error will be released to the market, however.
For general enquiries please contact Teva UK Customer Service Team on 0800 590502.
For medical information enquiries contact Teva UK limited by telephone on 0207 5407117 or by email to medinfo@tevauk.com.
Recipients of this drug alert should bring it to the attention of relevant contacts by copy of this letter. Local area teams are asked to forward this to relevant clinics, general practitioners and community pharmacists.
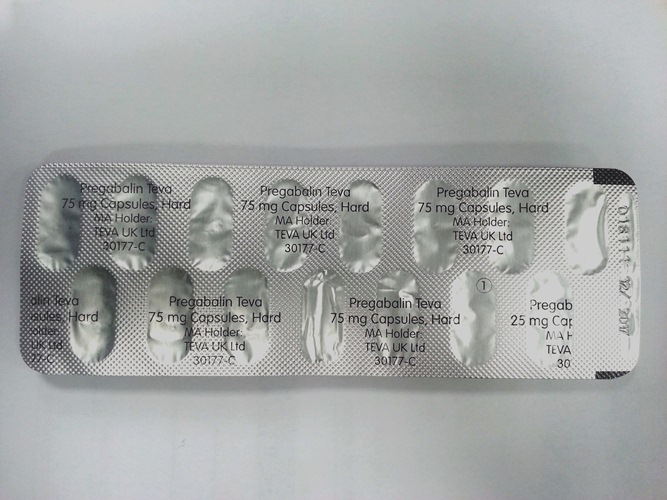
Image of Pregabalin 75mg Capsules.
Download documents