Class 4 Medicines Defect Notification: Doncaster Pharma Limited, Hiprex 1g tablets, EL(26)A/20
Doncaster Pharma Limited has informed the MHRA of an error related to the Braille embossing on the outer packaging of certain parallel imported batches of Hiprex 1 g tablets (POM).
DMRC reference number
DMRC- 39194203
Marketing Authorisation Holder
Doncaster Pharma Limited
Medicine Details
Hiprex 1g tablets
PLPI: 56830/0284
Active ingredient: methenamine hippurate
SNOMED code: 44366111000001107
GTIN: N/A
Affected Lot Batch Numbers
| Batch No. | Expiry Date | Pack Size | First Distributed |
|---|---|---|---|
| 251352/BA | 04/2028 | 100 tablets | 27/10/2025 |
| 251358/BA | 04/2028 | 100 tablets | 21/01/2026 |
| 252137/BA | 04/2028 | 100 tablets | 04/02/2026 |
| 252137/BB | 04/2028 | 100 tablets | 11/03/2026 |
| 251358/BB | 04/2028 | 100 tablets | 27/03/2026 |
Background
Doncaster Pharma Limited has informed the MHRA of an error related to the Braille embossing on the outer packaging of the above mentioned parallel imported batches of Hiprex 1 g tablets (POM). It was identified that the embossed Braille text on the outer packaging incorrectly states the strength as 1mg. The printed text on the outer packaging, immediate packaging and Patient Information Leaflet (PIL) correctly state the strength as 1g.
| Correct Braille | Incorrect Braille | |
|---|---|---|
| The correct Braille message should read: | The incorrect Braille message reads as: | |
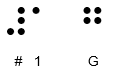 |
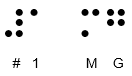 |
Advice for Healthcare Professionals:
The impacted products remain within specification and there is no issue with product quality. As a result, the affected batches are not being recalled.
Healthcare professionals should, if requested, explain the Braille errors to patients who rely solely on Braille when reading medicine cartons. As the products are supplied in a single strength, there is no risk of dosing errors arising from the Braille issue.
Advice for Patients:
No action is needed from patients, continue to take medication from these batches of tablets. The actions will be controlled by the healthcare professionals who prescribe or dispense the medication who will provide appropriate support at the time of dispensing, where required.
Patients who experience adverse reactions or have any questions about their medication should seek medical attention. Any suspected adverse reactions should also be reported via the MHRA Yellow Card scheme.
Additional information:
For all medical information enquiries and information on this product, please email regulatory@doncasterpharma.co.uk or quality.enquiries@doncasterpharma.co.uk, or telephone 01302 365 000.
For stock control enquiries, please email commercial@doncasterpharma.co.uk, or telephone 01302 365 000.
Recipients of this Medicines Notification should bring it to the attention of relevant contacts by copy of this notice. NHS regional teams are asked to forward this to community pharmacists and dispensing general practitioners for information.
Yours faithfully
Defective Medicines Report Centre
10 South Colonnade
Canary Wharf
London
E14 4PU
Telephone +44 (0)20 3080 6574
Download document