Class 2 Medicines Recall: Amarox Limited, Sertraline 100mg film-coated tablets, EL(26)A/22
Amarox Limited is recalling one batch of Sertraline 100 mg film-coated tablets as a precautionary measure due to an error at the manufacturing site.
DMRC reference number
DMRC - 39135670
Marketing Authorisation Holder
Amarox Limited
Medicine Details
Sertraline 100mg film-coated tablets
PL: 49445/0116
Active Ingredient: sertraline
SNOMED code: 41340311000001108
GTIN: 05060608561834
Affected Lot Batch Numbers
| Batch No. | Expiry Date | Pack Size | First Distributed |
|---|---|---|---|
| V2500425 | 05/2028 | 28 | 28 November 2025 |
Background
Amarox Limited is recalling one batch of Sertraline 100 mg film-coated tablets as a precautionary measure due to an error at the manufacturing site. Amarox Limited has received one complaint to date from an adult patient who reported the adverse event of headache, where it was identified that a pack of Sertraline 100 mg film-coated tablets (Batch Number V2500425) contains one blister strip of Citalopram 40 mg film-coated tablets inside the sealed carton. Both products were manufactured at the same manufacturing site and the error appears to have occurred during secondary packaging of the blister strips into the cartons.
Please note this is a Class 2 Patient, Pharmacy and Wholesaler level recall.
Advice for Healthcare Professionals:
Stop supplying the affected batch of Sertraline 100 mg film-coated tablets (Batch Number V2500425) immediately. Quarantine all remaining stock and return it to your supplier using your approved returns process.
-
If batch/product traceability information is available, pharmacy professionals and other healthcare professionals involved in dispensing should identify and immediately contact all patients who have been dispensed the impacted product and ask them to confirm if they have remaining stock within their possession for return.
-
If batch/product traceability information is not available, pharmacists should identify all patients dispensed this product from 28 November 2025 onwards. Where appropriate and feasible, contact all patients who have been dispensed the impacted product with priority given to those who have been dispensed the product most recently and within the last 28 days. The majority of this was distributed in 2025 and therefore it is expected that many of these packs will have been dispensed to patients and consumed.
-
If any patients are identified with this product, pharmacy professionals and other healthcare professionals involved in dispensing should contact the patients’ GP, or healthcare professional responsible for the care of the patient, to discuss treatment review and whether a new prescription is required for ongoing resupply.
Sertraline and citalopram are both selective serotonin reuptake inhibitors (SSRIs) used to treat depression, anxiety disorders, and related mental health conditions by boosting brain serotonin. Healthcare professionals should be advised to inform patients who may have inadvertently ingested citalopram tablets in place of or in addition to their prescribed sertraline, that they may experience increased serotonergic effects.
The reported adverse event in this case was a headache, which resolved on discontinuation; however, in susceptible individuals’ concurrent exposure to two SSRIs may carry a risk of more pronounced serotonergic effects. The risk is however higher in patients with pre-existing cardiac conditions, those aged over 65 years, or those taking concomitant serotonergic or QT prolonging medication.
Additionally, as some patients may require monitoring, including patients over 65 years old or under 18 years old, those with hepatic impairment and individuals who are known to be, or predicted to be a poor metaboliser for CYP2C19 substrates and therefore other clinicians and healthcare professionals may need to be involved. Healthcare professionals should be aware that only the batch stated in this notification is impacted.
Advice for Healthcare Professionals to Provide to Patients:
Some cartons of Sertraline 100 mg film-coated tablets, manufactured by Amarox Limited, may contain a blister strip(s) of Citalopram 40 mg tablets. All packs of Sertraline 100 mg film-coated tablets, Batch Number V2500425, are being recalled as a precautionary measure.
If you have been prescribed Sertraline 100 mg film-coated tablets and have received impacted product (Batch Number V2500425), please check that the carton contains the correct medication. The batch number and expiry date can be found printed on the side of the outer carton.
-
If the carton contains blister strips that are labelled and contain Citalopram 40 mg film-coated tablets, contact your dispensing pharmacy in the first instance.
-
If the carton contains blister strips that are labelled and contain Sertraline 100 mg film-coated tablets, you do not need to take further action.
If you have an impacted pack or previously received this batch, (where known) and you believe you have taken the Citalopram 40 mg film-coated tablets that were included in error and are currently experiencing any side effects, then please seek immediate medical advice.
Please take the leaflet that came with your medicine and any remaining tablets with you to your pharmacy or GP practice. If you are unsure or have any questions, please seek advice from your pharmacy or other healthcare professionals responsible for your care.
Both sertraline and citalopram are used to treat depression and anxiety; however, as your body may not be accustomed to citalopram, you may experience most common possible side effects such as nausea, headache, sleep changes and mild anxiety.
The description of the tablets from the Patient Information Leaflets (PILs) are as follows:
-
Sertraline 100 mg film-coated tablets: White to off white, film-coated, capsule shaped biconvex tablets and debossed with ‘3 and 1’ on either side of score line on one side and “HS” on other side. Click here for PIL
-
Citalopram 40 mg film-coated tablets: White to off-white coloured, film coated, oval biconvex tablets, debossed with “Z and 9” on either side of the score line and “H” on the other side. Click here for PIL
See below images showing the different medicines and where to find the batch number of the product and the identification of the incorrect blister strip.
Sertraline 100 mg film-coated tablets - Batch Number V2500425, Outer carton and Lot (Batch Number) /EXP Date information.
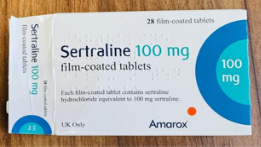

Citalopram 40 mg film-coated tablets – Batch Number V2500351 Incorrect blister strip

Patients who experience adverse reactions or have any questions about the medication should seek medical attention. Any suspected adverse reactions should also be reported via the MHRA Yellow Card scheme.
Additional information:
For all medical information enquiries, information and for reporting of side effects please email Safety@Amaroxpharma.com or telephone +44 2039720005 and for stock control enquiries please email info@amaroxpharma.com or telephone +44 2039720002.
Recipients of this Medicines Notification should bring it to the attention of relevant contacts by copy of this notice. NHS regional teams are asked to forward this to community pharmacists and dispensing general practitioners for information.
Yours faithfully
Defective Medicines Report Centre
10 South Colonnade
Canary Wharf
London
E14 4PU
Telephone +44 (0)20 3080 6574
Download document