Info on progressive multifocal leukoencephalopathy (PML) from Yellow Card reports
Spontaneous reporting on the Yellow Card portal has provided important information in the identification of PML associated with some drugs.
Article date: March 2009
The Yellow Card Scheme collects information on suspected adverse drug reactions in the UK.
Progressive multifocal leukoencephalopathy (PML) is a rare and usually fatal re-infection of the central nervous system, characterised by progressive damage and inflammation of the white matter in the brain, in multiple locations.
PML is caused by a type of human polyoma virus known as the JC, or John Cunningham virus. The JC virus is widespread, with about 70–90% of adults presenting antibodies.
The virus usually remains latent in healthy individuals, only causing disease when the immune system is severely compromised. PML has been studied in patients with HIV infection, where incidence is approximately 5% of the disease population. PML also occurs in patients with cancer and those who have received kidney or bone-marrow transplants.
In PML, gradual destruction of the myelin sheath covering nerve axons leads to impaired transmission of nerve impulses. PML causes rapidly progressive focal neurological deficits including: cognitive and behavioural changes; paraesthesia; visual problems; gait abnormalities and loss of limb coordination; and hemiparesis.
Drugs associated with PML
MHRA has previously identified an association between PML and use of some monoclonal antibodies such as natalizumab (Tysabri ▼, used to treat multiple sclerosis) and rituximab (MabThera, indicated for non-Hodgkin’s lymphoma and severe active rheumatoid arthritis). An association has also been identified between PML and efalizumab (Raptiva ▼, a treatment for moderate to severe plaque psoriasis). A European review has concluded that the benefits of this drug do not outweigh its risks, and the European Medicines Agency has recommended the suspension of the marketing authorisation for efalizumab.
Yellow Card reports for PML
Up to Jan 6 2009, the MHRA has received 19 suspected reports of PML, 3 of which where PML was listed as the fatal suspected reaction.
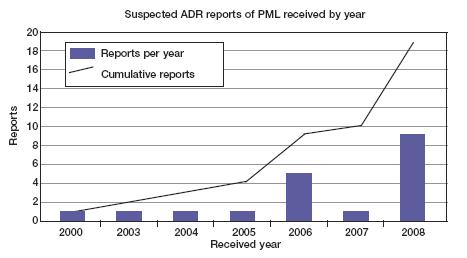
Image of graph showing suspected ADR reports.
The table lists the drugs most reported to the MHRA in association with suspected PML:
| Suspected drug | Number of cases PML* | |
|---|---|---|
| Cyclophosphamide (antineoplastic agent) | 5 | |
| Rituximab (monoclonal antibody) | 5 | |
| Epirubicin (antineoplastic agent) | 3 | |
| Fludarabine (antineoplastic agent) | 3 |
*Note that more than 1 drug may be implicated in a case of PML.
Warnings about PML are given in the product information for rituximab, alemtuzumab, natalizumab, fludarabine, nelarabine, and mycophenolate mofetil. There is currently insufficient evidence of a causal relation between cyclophosphamide or epirubicin and PML, and PML is not currently listed in the product safety information for these drugs. The risk of drug-induced PML continues to be monitored closely by MHRA.
Report suspected reactions of PML
Spontaneous reporting has provided important information in the identification of PML associated with some drugs. If you suspect the involvement of a drug in a case of PML, please let us know by filling in a Yellow Card. We are particularly interested in the time to onset of PML after starting treatment, indications for suspected drugs, and all other medicines the patient is receiving.
If in doubt, report.
Article citation: Drug Safety Update March 2009, vol 2 issue 8: 9.