Bisphosphonates: osteonecrosis of the jaw
All patients with cancer should have a dental check-up before bisphosphonate treatment.
Article date: November 2009
Individual bisphosphonates have different indications, and are used for: prophylaxis and treatment of osteoporosis; treatment of Paget’s disease; and as part of some cancer regimens, particularly for metastatic bone cancer and multiple myeloma.
A Europe-wide review has been completed on the risk of osteonecrosis of the jaw (ONJ) in association with the use of bisphosphonates. The review included data from the published literature and data provided by the Marketing Authorisation Holders (including data from experimental and preclinical studies, clinical trials, and post-marketing reports) and guidelines produced by learned societies. The review also incorporated advice from a group of experts representing all areas of medicine where bisphosphonates are used, dentistry and bone surgery, and representatives of patients’ organisations.
Up to Oct 26, 2009, the majority of UK Yellow Card reports of ONJ have been in patients treated with zoledronate (172 reports). Yellow Card reports of ONJ have also been received in association with alendronate (47 reports), pamidronate (38 reports), ibandronate (28 reports), clodronate (12 reports), and risedronate (11 reports). It is very important to remember that the number of reports for a particular bisphosphonate cannot be used to determine the incidence of ONJ for each individual drug because neither the total number of reactions occurring nor the number of patients using the drug is known.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) reached conclusions on the currently available evidence on four main areas: definition and diagnosis of ONJ related to bisphosphonates; possible underlying pathophysiological mechanism(s); risk stratification; and risk minimisation. The key messages for healthcare professionals from this review are given below.
Advice for healthcare professionals:
- The risk of developing ONJ in association with oral bisphosphonates seems to be low. The risk of ONJ is substantially greater for patients receiving intravenous bisphosphonates for cancer indications than for patients receiving oral bisphosphonates for osteoporosis or Paget’s disease
- There is clear evidence to suggest bisphosphonate-specific and indication-specific risk factors such as potency (highest for zoledronate); route of administration (eg, intravenous ibandronte, pamidronate, and zoledronate); and cumulative dose. The evidence base is less robust for other proposed risk factors (eg, duration and type of malignant disease,concomitant treatment, smoking, and comorbid conditions). However, healthcare professionals should consider these risk factors when evaluating an individual’s risk of developing ONJ
- A history of dental disease—including invasive dental procedures, dental trauma, periodontal disease, and poorly fitting dentures—is associated with an increased risk of ONJ
Risk minimisation:
- All patients with cancer should have a dental check-up before bisphosphonate treatment. All other patients who start bisphosphonates should have a dental examination only if they have poor dental status
- During bisphosphonate treatment, patients should maintain good oral hygiene, receive routine dental check-ups, and report any oral symptoms such as dental mobility, pain, or swelling
The risk of ONJ with bisphosphonates will be kept under close review in Europe. Further research is needed to increase knowledge about the underlying mechanisms and risk factors for ONJ, and about how best to minimise these risks. The EU regulatory authorities will explore strategies to promote this research, and the CHMP has adopted a definition (see margin note, p 2) of ONJ related to bisphosphonates to facilitate future case reporting and research.
A patient may be considered to have ONJ related to bisphosphonates if all of the following three characteristics are present:
- Exposed or necrotic bone in the maxillofacial region that has persisted for more than 8 weeks
- No history of irradiation of the jaw
- Current or previous treatment with a bisphosphonate
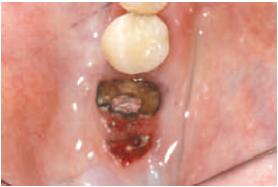
Image of Osteonecrosis of the jaw.
Osteonecrosis of the jaw
(Image credit: John W Hellstein, University of Iowa College of Dentistry and Hardin MD, University of Iowa.)
Further information is available from the EMEA
Article citation: Drug Safety Update Nov 2009, vol 3 issue 4: 2.